
Genome Editing Market
Genome Editing Market Forecasts to 2028 - Global Analysis By Product & Service (Reagents & Consumables, Services, Software & Systems, Cell Lines and Other Product & Services), By Technology (Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR), Zinc Finger Nuclease (ZFN), Transcription Activator-Like Effector Nuclease (TALEN), Antisense, Restriction Enzymes and Other Technologies), By Application (Cell Line Editing, Genetic Engineering, Drug Discovery & Development, Diagnostic Applications, Microorganisms Genetic Engineering, Gene Therapy and Other Applications), By End User (Clinical Research Organizations, Academics & Government Research Institutes, Pharmaceutical & Biotechnology Companies, Contract Research Organization and Other End Users) and By Geography

|
Years Covered |
2020-2028 |
|
Estimated Year Value (2022) |
US $6.1 BN |
|
Projected Year Value (2028) |
US $18.5 BN |
|
CAGR (2022 - 2028) |
20.2% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
North America |
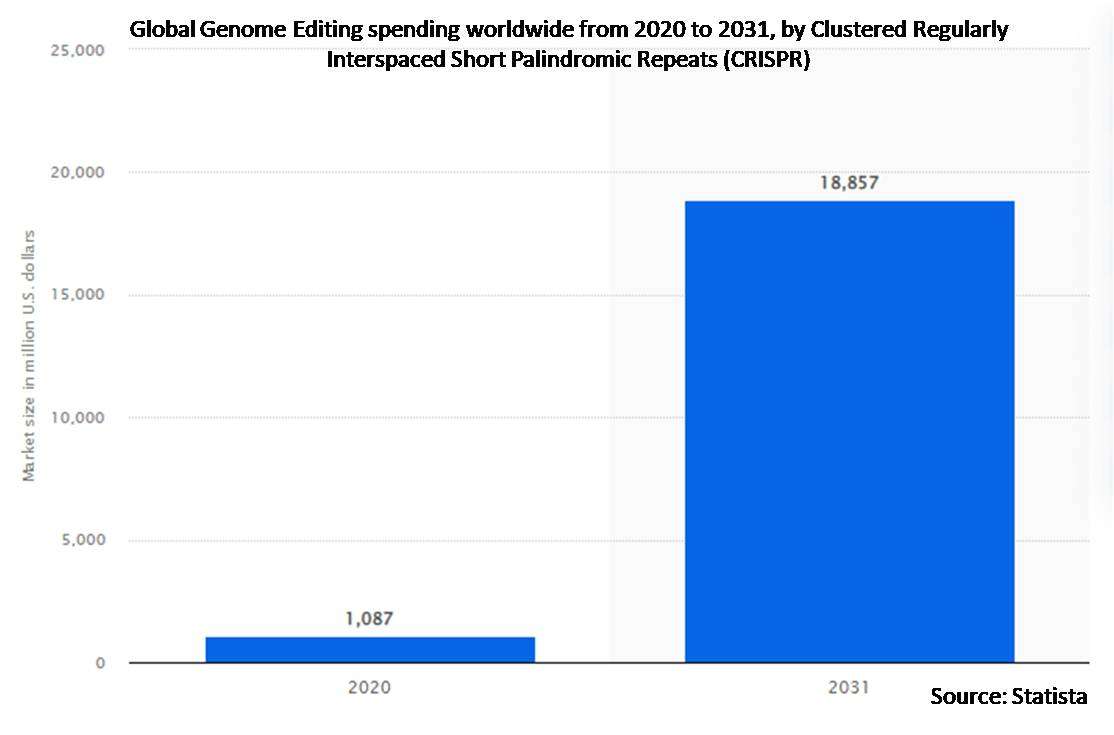
According to Stratistics MRC, the Global Genome Editing Market is accounted for $6.1 billion in 2022 and is expected to reach $18.5 billion by 2028 growing at a CAGR of 20.2% during the forecast period. Gene editing is the use of instruments and processes that significantly change the genetic blueprints of a living thing by making extremely particular alterations to the DNA sequence of a gene. Gene editing strives to modify particular target regions, as opposed to genetic engineering, which randomly combines genetic material into a host genome.
According to the National Center for Advancing Translational Sciences updates form June 2022, the Somatic Cell Genome Editing (SCGE) Program at the National Institutes of Health (NIH) has awarded 24 more grants to researchers across the United States and Canada.
Market Dynamics:
Driver:
Developments in genome editing
Recent developments have substantially improved genome editing capabilities, enabling the alteration of unique gene sequences in a variety of cell types and model species. In particular, for oncology tests, genome engineering has developed into a potentially game-changing diagnostic tool. Genome engineering was previously used by major research institutions, including those affiliated with the government and universities. This significant movement away from academics and into clinical and diagnostic facilities is primarily driven by the rapid advancement of gene editing technology.
Restraint:
High equipment costs
Because genomic instruments have cutting-edge features and functions, they are more expensive, with costs averaging between USD 10 and USD 20 million. As many of these systems are needed by pharmaceutical businesses and research labs, their capital expenditure to acquire several genomic equipment is very significant. It is not practical for small and medium-sized pharmaceutical corporations and research labs to invest substantially in numerous genomics devices.
Opportunity:
Extensive use of genome editing in personalized medicine
Players in the genome editing market should have opportunities as the field of customized medicine grows. With the aid of personalized medicine, practitioners may identify a patient's precise needs and then prescribe medications and treatments in response. Due to its potential for cost reductions, better patient compliance, early disease identification, and therapy improvement, the use of customized medicine is expanding globally.
Threat:
Off-target effects of CRISPR technology
CRISPR-Cas9's off-target consequences are a significant concern. Any off-target nuclease activity may modify these genes and result in oncogenesis since Cas9 generates double-stranded breaks. One to three mismatches in the target can be tolerated by CRISPR-Cas9, which can result in off-target nuclease activity. A significant concern is the high frequency of off-target activity (50%), which refers to alterations at locations other than the planned on-target site. For instance, CRISPR can activate the cancer-causing gene or target the tumor suppressor gene. Due to this undesirable effect, numerous companies that are organizing clinical studies have experienced problems. Clinical trials have been revoked, and regulatory authorities demand more studies to improve the safety of this procedure.
Covid-19 Impact:
COVID-19 has a favorable effect on the market for genome editing. The COVID-19 epidemic has increased the strain on healthcare systems around the world. Major pharmaceutical and biotechnology companies have increased their investments in vaccine R&D and market players for genome editing as a result of the COVID-19 pandemic. Due to the rising demand for vaccinations and potential antiviral medications, genome editing and engineering technologies are at the forefront of scientific research. Additionally, there has been a moderate impact on the market for genome editing as well as other healthcare services. Many researchers switched their attention to the investigation of this unique virus and the illness it produces after COVID-19 was originally identified.
The clustered regularly interspaced short palindromic repeats (CRISPR) segment is expected to be the largest during the forecast period
Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) segment is expected to have moderate growth over the projected period because of increased government financing and initiatives to develop vaccinations, medical technology, pharmaceuticals, and gadgets. In two cases of inherited diseases—one in a patient with TDT (thalassemia) and the other in a patient with SCD (sickle cell disease)—CRISPR-Cas9-based gene editing is being tested, according to a study titled "CRISPR-Cas9 Gene Editing for Sickle Cell Disease and -Thalassemia," published in the New England Journal of Medicine in January 2021. Both patients witnessed early, significant, and long-lasting increases in fetal hemoglobin levels with more than 99% pancellularity throughout the course of the following year after receiving CTX001. Therefore, it is anticipated that over the forecast period, demand for CRISPR technology will increase due to its use in treating diseases including sickle cell disease and beta-thalassemia.
The cell line editing segment is expected to have the highest CAGR during the forecast period
Due to the expanding use of cell line engineering in stem cell-based research, it is predicted that the cell line editing segment will have profitable growth over an extended period. Cell line engineering is widely used for induced pluripotent stem cells (iPSCs), which help to change the genetic material of the cells as a result of rapid improvements. Additionally, advanced gene editing methods, including zinc finger nucleases (ZFNs), CRISPR/Cas, and TALENs, have made cell editing affordable and feasible.
Region with largest share:
Due to the biotechnology and pharmaceutical industries' strong development trends, North America has acquired a monopoly on the global market for genome editing. During the anticipated period, it is predicted that factors including technological advancements in genome editing, rising product approvals, and expanding R&D procedures would accelerate market expansion. However, the United States Food and Drug Administration provided consent to researchers at the University of California (UC), San Francisco, UC Berkeley, and UC Los Angeles in March 2021 to jointly begin an early-phase, first-in-human clinical trial of a gene correction therapy in sickle cell disease patients using the patient's blood-forming stem cells.
Region with highest CAGR:
Due to the advancement of gene therapy, growing consumption of genetically modified crops, and easy access to research funding, North America hold the highest CAGR over the forecasted period and will continue to make the largest contribution to businesses offering tools and services for gene editing. Additionally, the U.S. controls the majority of the market in North America, mostly as a result of increased public and private financing for genomics research. Furthermore, the market for genome editing in North America is being driven by the rising prevalence of rare illnesses in the United States and Canada. More than 7,000 rare diseases affect between 25 and 30 million Americans, according to the National Institutes of Health (NIH). According to this statistic, one in ten Americans has a rare disorder. As a result, the adoption of novel DNA editing methods in these nations is driven by the growing need to provide effective medications.
Key players in the market
Some of the key players in Genome Editing market include Agilent Technologies Inc., AstraZeneca, Beam Therapeutics, Bluebird Bio, Caribou Biosciences, Inc, Cellectis S.A., Cibus, Recombinetics, Inc., CRISPR Therapeutics AG, Editas Medicine Inc., Egenesis Inc., GenScript Biotech Corporation, GenScript USA Inc., Horizon Discovery Group plc, Integrated DNA Technologies Inc., Intellia Therapeutics, Inc., Lonza Group Ltd, Merck KGaA, New England Biolabs Inc., Origene Technologies Inc., Precision BioSciences Inc., Sangamo Therapeutics Inc., Takara Bio Inc., Tecan Life Sciences , Thermo Fisher Scientific Inc. and Transposagen Biopharmaceuticals Inc.
Key Developments:
In May 2022, the English parliament introduced a Bill that would enable the release of genetically edited products. The Genetic Technology (Precision Breeding) Bill aims to benefit companies, farmers, and consumers. The bill is currently under the report stage in the House of Commons.
In February 2022, Integrated DNA Technologies unveiled Alt-R HDR Donor Blocks, an improved solution for increasing homology-directed repair (HDR) rates. Alt-R HDR Donor Blocks are the latest addition to IDT’s complete portfolio of Alt-R CRISPR genome editing solutions.
In February 2022, Intellia Therapeutics, Inc. and Regeneron Pharmaceuticals, Inc. announced the positive interim data from an ongoing Phase I clinical study of their lead in vivo genome editing candidate, NTLA-2001 (Investigational Crispr Therapy For Transthyretin (Attr) Amyloidosis).
In January 2022, CRISPR Therapeutics AG announced a strategic partnership with Capsida Biotherapeutics Inc. to develop new gene therapies using the CRISPR/Cas9 technology for the treatment of various diseases. Under the agreement, CRISPR Therapeutics will provide its genome editing expertise, while Capsida will contribute its adeno-associated virus (AAV) delivery technology.
Product & Services Covered:
• Reagents & Consumables
• Services
• Software & Systems
• Cell Lines
• Other Product & Services
Technologies Covered:
• Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)
• Zinc Finger Nuclease (ZFN)
• Transcription Activator-Like Effector Nuclease (TALEN)
• Antisense
• Restriction Enzymes
• Other Technologies
Applications Covered:
• Cell Line Editing
• Genetic Engineering
• Drug Discovery & Development
• Diagnostic Applications
• Microorganisms Genetic Engineering
• Gene Therapy
• Other Applications
End Users Covered:
• Clinical Research Organizations
• Academics & Government Research Institutes
• Pharmaceutical & Biotechnology Companies
• Contract Research Organization
• Other End Users
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2020, 2021, 2022, 2025, and 2028
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Technology Analysis
3.7 Application Analysis
3.8 End User Analysis
3.9 Emerging Markets
3.10 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Genome Editing Market, By Product & Service
5.1 Introduction
5.2 Reagents & Consumables
5.3 Services
5.4 Software & Systems
5.5 Cell Lines
5.6 Other Product & Services
6 Global Genome Editing Market, By Technology
6.1 Introduction
6.2 Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)
6.3 Zinc Finger Nuclease (ZFN)
6.4 Transcription Activator-Like Effector Nuclease (TALEN)
6.5 Antisense
6.6 Restriction Enzymes
6.7 Other Technologies
7 Global Genome Editing Market, By Application
7.1 Introduction
7.2 Cell Line Editing
7.3 Genetic Engineering
7.3.1 Plant Genetic Engineering
7.3.2 Animal Genetic Engineering
7.4 Drug Discovery & Development
7.5 Diagnostic Applications
7.6 Microorganisms Genetic Engineering
7.7 Gene Therapy
7.8 Other Applications
8 Global Genome Editing Market, By End User
8.1 Introduction
8.2 Clinical Research Organizations
8.3 Academics & Government Research Institutes
8.4 Pharmaceutical & Biotechnology Companies
8.5 Contract Research Organization
8.6 Other End Users
9 Global Genome Editing Market, By Geography
9.1 Introduction
9.2 North America
9.2.1 US
9.2.2 Canada
9.2.3 Mexico
9.3 Europe
9.3.1 Germany
9.3.2 UK
9.3.3 Italy
9.3.4 France
9.3.5 Spain
9.3.6 Rest of Europe
9.4 Asia Pacific
9.4.1 Japan
9.4.2 China
9.4.3 India
9.4.4 Australia
9.4.5 New Zealand
9.4.6 South Korea
9.4.7 Rest of Asia Pacific
9.5 South America
9.5.1 Argentina
9.5.2 Brazil
9.5.3 Chile
9.5.4 Rest of South America
9.6 Middle East & Africa
9.6.1 Saudi Arabia
9.6.2 UAE
9.6.3 Qatar
9.6.4 South Africa
9.6.5 Rest of Middle East & Africa
10 Key Developments
10.1 Agreements, Partnerships, Collaborations and Joint Ventures
10.2 Acquisitions & Mergers
10.3 New Product Launch
10.4 Expansions
10.5 Other Key Strategies
11 Company Profiling
11.1 Agilent Technologies Inc.
11.2 AstraZeneca
11.3 Beam Therapeutics
11.4 Bluebird Bio
11.5 Caribou Biosciences, Inc
11.6 Cellectis S.A.
11.7 Cibus, Recombinetics, Inc.
11.8 CRISPR Therapeutics AG
11.9 Editas Medicine Inc.
11.10 Egenesis Inc.
11.11 GenScript Biotech Corporation
11.12 GenScript USA Inc.
11.13 Horizon Discovery Group plc
11.14 Integrated DNA Technologies Inc.
11.15 Intellia Therapeutics, Inc.
11.16 Lonza Group Ltd
11.17 Merck KGaA
11.18 New England Biolabs Inc.
11.19 Origene Technologies Inc.
11.20 Precision BioSciences Inc.
11.21 Sangamo Therapeutics Inc.
11.22 Takara Bio Inc.
11.23 Tecan Life Sciences
11.24 Thermo Fisher Scientific Inc.
11.25 Transposagen Biopharmaceuticals Inc.
List of Tables
1 Global Genome Editing Market Outlook, By Region (2020-2028) ($MN)
2 Global Genome Editing Market Outlook, By Product & Service (2020-2028) ($MN)
3 Global Genome Editing Market Outlook, By Reagents & Consumables (2020-2028) ($MN)
4 Global Genome Editing Market Outlook, By Services (2020-2028) ($MN)
5 Global Genome Editing Market Outlook, By Software & Systems (2020-2028) ($MN)
6 Global Genome Editing Market Outlook, By Cell Lines (2020-2028) ($MN)
7 Global Genome Editing Market Outlook, By Other Product & Services (2020-2028) ($MN)
8 Global Genome Editing Market Outlook, By Technology (2020-2028) ($MN)
9 Global Genome Editing Market Outlook, By Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) (2020-2028) ($MN)
10 Global Genome Editing Market Outlook, By Zinc Finger Nuclease (ZFN) (2020-2028) ($MN)
11 Global Genome Editing Market Outlook, By Transcription Activator-Like Effector Nuclease (TALEN) (2020-2028) ($MN)
12 Global Genome Editing Market Outlook, By Antisense (2020-2028) ($MN)
13 Global Genome Editing Market Outlook, By Restriction Enzymes (2020-2028) ($MN)
14 Global Genome Editing Market Outlook, By Other Technologies (2020-2028) ($MN)
15 Global Genome Editing Market Outlook, By Application (2020-2028) ($MN)
16 Global Genome Editing Market Outlook, By Cell Line Editing (2020-2028) ($MN)
17 Global Genome Editing Market Outlook, By Genetic Engineering (2020-2028) ($MN)
18 Global Genome Editing Market Outlook, By Plant Genetic Engineering (2020-2028) ($MN)
19 Global Genome Editing Market Outlook, By Animal Genetic Engineering (2020-2028) ($MN)
20 Global Genome Editing Market Outlook, By Drug Discovery & Development (2020-2028) ($MN)
21 Global Genome Editing Market Outlook, By Diagnostic Applications (2020-2028) ($MN)
22 Global Genome Editing Market Outlook, By Microorganisms Genetic Engineering (2020-2028) ($MN)
23 Global Genome Editing Market Outlook, By Gene Therapy (2020-2028) ($MN)
24 Global Genome Editing Market Outlook, By Other Applications (2020-2028) ($MN)
25 Global Genome Editing Market Outlook, By End User (2020-2028) ($MN)
26 Global Genome Editing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
27 Global Genome Editing Market Outlook, By Academics & Government Research Institutes (2020-2028) ($MN)
28 Global Genome Editing Market Outlook, By Pharmaceutical & Biotechnology Companies (2020-2028) ($MN)
29 Global Genome Editing Market Outlook, By Contract Research Organization (2020-2028) ($MN)
30 Global Genome Editing Market Outlook, By Other End Users (2020-2028) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We offer complimentary customization of up to 15% with every purchase. To share your customization requirements, feel free to email us at info@strategymrc.com or call us on +1-301-202-5929. .
Please Note: Customization within the 15% threshold is entirely free of charge. If your request exceeds this limit, we will conduct a feasibility assessment. Following that, a detailed quote and timeline will be provided.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
