
Sterile Injectable Market
Sterile Injectable Market Forecasts to 2030 - Global Analysis By Drug Type (Cytokines, Monoclonal Antibodies, Insulin, Peptide Hormones, Blood Factors, Vaccines, Immunoglobulins, Peptide Antibiotics and Other Drug Types), Molecule Type, Route of Administration, Distribution Channel, Application and By Geography

|
Years Covered |
2021-2030 |
|
Estimated Year Value (2023) |
US $62.1 BN |
|
Projected Year Value (2030) |
US $115.0 BN |
|
CAGR (2023 - 2030) |
9.2% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
Asia Pacific |
According to Stratistics MRC, the Global Sterile Injectable Market is accounted for $62.1 billion in 2023 and is expected to reach $115.0 billion by 2030 growing at a CAGR of 9.2% during the forecast period. Pharmaceuticals that are given intravenously and must be devoid of live germs like bacteria or fungus are referred to as sterile injectables. These injectables can be given intravenously (IV), intramuscularly (IM), or subcutaneously (SC). They are designed to be directly injected into the circulation or other sterile bodily tissues. To guarantee that there is no microbiological contamination in the finished product, they are usually prepared under stringent aseptic processing conditions.
According to the National Health Council, in 2016, chronic diseases affect approximately 133 million Americans, representing more than 40% of the total population of the country. By 2020, that number is projected to grow to an estimated 157 million, with 81 million having multiple conditions.
Market Dynamics:
Driver:
Rise in inclination towards sterile injectables
Sterile injectables, such as medicines, biologics, and vaccinations, are essential to healthcare, particularly for managing diseases and in emergency circumstances. New treatment methods, many of which are injected, have been discovered and developed as a result of advancements in medical research. This covers novel medical interventions such as gene and cell therapy. These are the aspects driving market growth throughout the predicted period.
Restraint:
Risk of contamination
Although sterility is maintained to the best of our ability, contamination can still occur during production, packing, or administration. For patients, contaminants can result in major health problems. Additionally, in order to preserve stability, some injectable medications may need special storage conditions, such refrigeration. Creating additional logistical hurdles for transportation and storage, particularly in areas with poor access to adequate facilities restrains the market growth.
Opportunity:
Increased bioavailability
Sterile injectables frequently offer better bioavailability than oral drugs because they are not susceptible to the unpredictability of gastrointestinal absorption. Medications that are injectable frequently start working faster than those that are oral. For drugs with a limited therapeutic window or those needing a certain dosage for best efficacy, they are beneficial since they deliver larger drug concentrations straight into the circulation. During the anticipated time, these factors will be propelling market expansion.
Threat:
Packaging concerns
Sterilized injectable packaging is essential to preserving the integrity of the product. A breach in the package may result in contamination or a reduction in the medication's effectiveness. Furthermore, the production and quality assurance procedures for sterile injectables can be expensive and complicated. Higher expenses follow for people, healthcare systems, and pharmaceutical corporations alike. Thus, these are the things impeding the market's expansion.
Covid-19 Impact:
The pandemic caused supply chain disruptions on a worldwide scale, resulting in shortages of raw materials, active pharmaceutical ingredients (APIs), and packaging supplies. This has an impact on the distribution and manufacturing of sterile injectables. Manufacturing activities were disrupted by personnel constraints, social distancing measures, and lockdowns. Due to workforce shortages and mobility constraints, several pharmaceutical businesses found it difficult to sustain production levels.
The insulin segment is expected to be the largest during the forecast period
The insulin segment is expected to be the largest during the forecast period. Insulin that is sterile for injection is an essential part of managing diabetes. By assisting in blood glucose regulation, it helps avoid hypo- and hyperglycemia. With injectable insulin, dosage may be precisely and uniquely adjusted for each patient depending on their unique requirements, lifestyle, and blood glucose levels. To customize therapy, a variety of insulin types with different onset and duration characteristics are available.
The cancer segment is expected to have the highest CAGR during the forecast period
The cancer segment is expected to have the highest CAGR during the forecast period. Medication may be administered precisely and under control to sterile injectables. This is important because, in the treatment of cancer, precise dosage is necessary to maximize therapeutic efficacy and reduce adverse effects. This can be especially crucial for cancer therapy, as it may be required to act quickly to stop the growth and spread of cancer cells.
Region with largest share:
North America is projected to hold the largest market share during the forecast period due to the rise in prevalence of chronic diseases and disorders among population. The region's well-developed healthcare infrastructure, well-known companies, attractive reimbursement rules, and strong use in the cancer sector are all credited with this expansion. Furthermore, regulating organizations like the Food and Drug Administration (FDA) of the United States are essential in guaranteeing the security and caliber of sterile injectable medications in the area.
Region with highest CAGR:
Asia Pacific is projected to hold the highest CAGR over the forecast period due to the increasing prevalence of cancer cases among people and surging investment by government with the purpose of enhancing healthcare sector. In healthcare settings, sterile injectables are essential for administering a range of treatments, such as specialized medications, vaccinations, and antibiotics. Sterile injectable medications are often manufactured and distributed in the region by major pharmaceutical businesses, both domestic and foreign.
Key players in the market
Some of the key players in Sterile Injectable market include Novartis AG, Baxter International Inc., Merck & Co., Inc, AstraZeneca plc, Johnson & Johnson Services, Inc, Gilead Sciences, Inc., JHP Pharmaceuticals, Pfizer Inc., Fresenius Kabi Ag, CordenPharma, Hikma Pharmaceuticals PLC, Bryllon LLC, Aenova Holding GmbH, Vetter Pharma-Fertigung GmbH & Co. KG and Catalent Pharma Solutions.
Key Developments:
In November 2023, AstraZeneca announced a collaboration and investment agreement with Cellectis, a clinical-stage biotechnology company, to accelerate the development of next generation therapeutics in areas of high unmet need, including oncology, immunology and rare diseases.
In January 2022, Johnson & Johnson Medical Devices Companies announced that it will collaborate with Microsoft to further enable and expand JJMDC’s secure and compliant digital surgery ecosystem.
In January 2021, Novartis announced collaboration with Alnylam to leverage Alnylam’s proven, proprietary siRNA technology to inhibit a target discovered at the Novartis Institutes for BioMedical Research, potentially leading to development of a treatment designed to promote the regrowth of functional liver cells and to provide an alternative to transplantation for patients with liver failure.
Drug Types Covered:
• Cytokines
• Monoclonal Antibodies
• Insulin
• Peptide Hormones
• Blood Factors
• Vaccines
• Immunoglobulins
• Peptide Antibiotics
• Other Drug Types
Molecule Types Covered:
• Large Molecules
• Small Molecules
Route of Administrations Covered:
• Intravenous (IV)
• Intramuscular (IM)
• Subcutaneous (SC)
Distribution Channels Covered:
• Hospital Pharmacies
• Retail Pharmacies
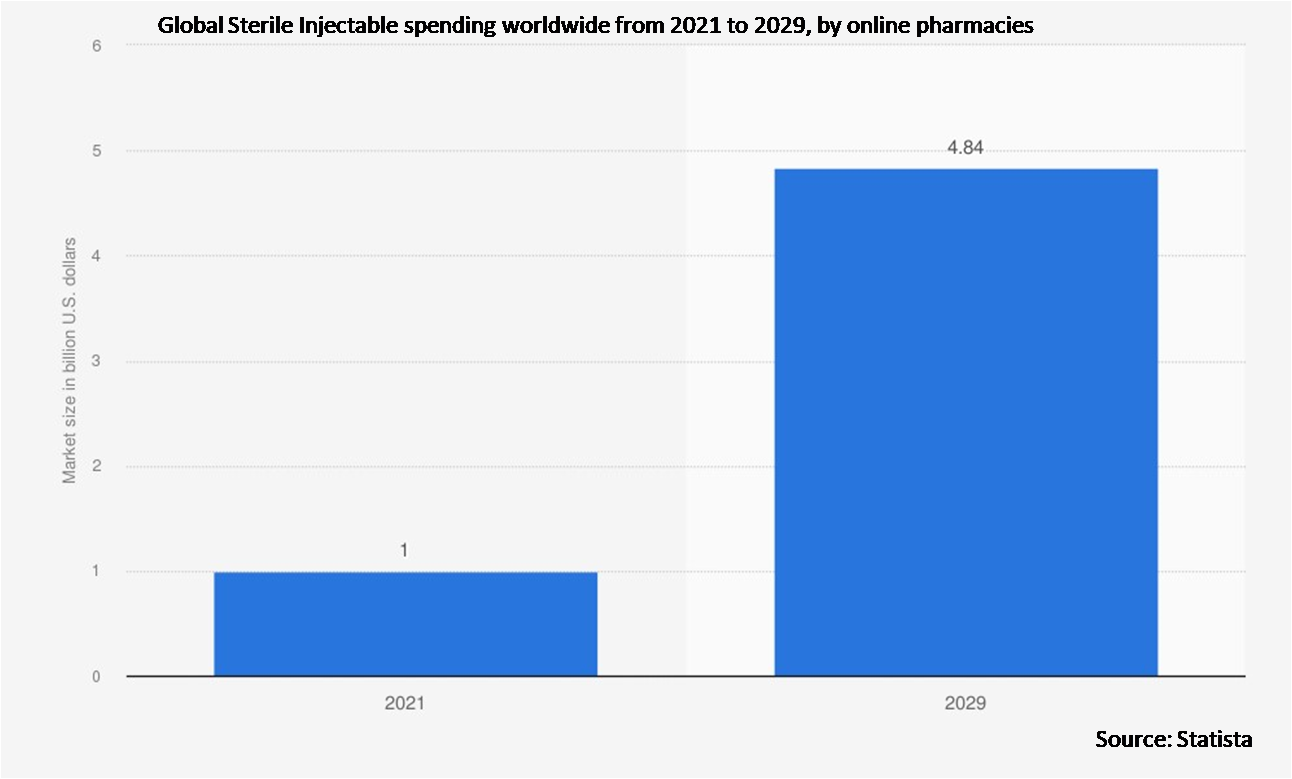
• Online Pharmacies
Applications Covered:
• Cardiovascular Diseases
• Musculoskeletal
• Cancer
• Infections
• Diabetes
• Central Nervous Systems
• Other Applications
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2021, 2022, 2023, 2026, and 2030
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
o Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Application Analysis
3.7 Emerging Markets
3.8 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Sterile Injectable Market, By Drug Type
5.1 Introduction
5.2 Cytokines
5.3 Monoclonal Antibodies
5.4 Insulin
5.5 Peptide Hormones
5.6 Blood Factors
5.7 Vaccines
5.8 Immunoglobulins
5.9 Peptide Antibiotics
5.10 Other Drug Types
6 Global Sterile Injectable Market, By Molecule Type
6.1 Introduction
6.2 Large Molecules
6.3 Small Molecules
7 Global Sterile Injectable Market, By Route of Administration
7.1 Introduction
7.2 Intravenous (IV)
7.3 Intramuscular (IM)
7.4 Subcutaneous (SC)
8 Global Sterile Injectable Market, By Distribution Channel
8.1 Introduction
8.2 Hospital Pharmacies
8.3 Retail Pharmacies
8.4 Online Pharmacies
9 Global Sterile Injectable Market, By Application
9.1 Introduction
9.2 Cardiovascular Diseases
9.3 Musculoskeletal
9.4 Cancer
9.5 Infections
9.6 Diabetes
9.7 Central Nervous Systems
9.8 Other Applications
10 Global Sterile Injectable Market, By Geography
10.1 Introduction
10.2 North America
10.2.1 US
10.2.2 Canada
10.2.3 Mexico
10.3 Europe
10.3.1 Germany
10.3.2 UK
10.3.3 Italy
10.3.4 France
10.3.5 Spain
10.3.6 Rest of Europe
10.4 Asia Pacific
10.4.1 Japan
10.4.2 China
10.4.3 India
10.4.4 Australia
10.4.5 New Zealand
10.4.6 South Korea
10.4.7 Rest of Asia Pacific
10.5 South America
10.5.1 Argentina
10.5.2 Brazil
10.5.3 Chile
10.5.4 Rest of South America
10.6 Middle East & Africa
10.6.1 Saudi Arabia
10.6.2 UAE
10.6.3 Qatar
10.6.4 South Africa
10.6.5 Rest of Middle East & Africa
11 Key Developments
11.1 Agreements, Partnerships, Collaborations and Joint Ventures
11.2 Acquisitions & Mergers
11.3 New Product Launch
11.4 Expansions
11.5 Other Key Strategies
12 Company Profiling
12.1 Novartis AG
12.2 Baxter International Inc.
12.3 Merck & Co., Inc
12.4 AstraZeneca plc
12.5 Johnson & Johnson Services, Inc
12.6 Gilead Sciences, Inc.
12.7 JHP Pharmaceuticals
12.8 Pfizer Inc.
12.9 Fresenius Kabi Ag
12.10 CordenPharma
12.11 Hikma Pharmaceuticals PLC
12.12 Bryllon LLC
12.13 Aenova Holding GmbH
12.14 Vetter Pharma-Fertigung GmbH & Co. KG
12.15 Catalent Pharma Solutions
List of Tables
1 Global Sterile Injectable Market Outlook, By Region (2021-2030) ($MN)
2 Global Sterile Injectable Market Outlook, By Drug Type (2021-2030) ($MN)
3 Global Sterile Injectable Market Outlook, By Cytokines (2021-2030) ($MN)
4 Global Sterile Injectable Market Outlook, By Monoclonal Antibodies (2021-2030) ($MN)
5 Global Sterile Injectable Market Outlook, By Insulin (2021-2030) ($MN)
6 Global Sterile Injectable Market Outlook, By Peptide Hormones (2021-2030) ($MN)
7 Global Sterile Injectable Market Outlook, By Blood Factors (2021-2030) ($MN)
8 Global Sterile Injectable Market Outlook, By Vaccines (2021-2030) ($MN)
9 Global Sterile Injectable Market Outlook, By Immunoglobulins (2021-2030) ($MN)
10 Global Sterile Injectable Market Outlook, By Peptide Antibiotics (2021-2030) ($MN)
11 Global Sterile Injectable Market Outlook, By Other Drug Types (2021-2030) ($MN)
12 Global Sterile Injectable Market Outlook, By Molecule Type (2021-2030) ($MN)
13 Global Sterile Injectable Market Outlook, By Large Molecules (2021-2030) ($MN)
14 Global Sterile Injectable Market Outlook, By Small Molecules (2021-2030) ($MN)
15 Global Sterile Injectable Market Outlook, By Route of Administration (2021-2030) ($MN)
16 Global Sterile Injectable Market Outlook, By Intravenous (IV) (2021-2030) ($MN)
17 Global Sterile Injectable Market Outlook, By Intramuscular (IM) (2021-2030) ($MN)
18 Global Sterile Injectable Market Outlook, By Subcutaneous (SC) (2021-2030) ($MN)
19 Global Sterile Injectable Market Outlook, By Distribution Channel (2021-2030) ($MN)
20 Global Sterile Injectable Market Outlook, By Hospital Pharmacies (2021-2030) ($MN)
21 Global Sterile Injectable Market Outlook, By Retail Pharmacies (2021-2030) ($MN)
22 Global Sterile Injectable Market Outlook, By Online Pharmacies (2021-2030) ($MN)
23 Global Sterile Injectable Market Outlook, By Application (2021-2030) ($MN)
24 Global Sterile Injectable Market Outlook, By Cardiovascular Diseases (2021-2030) ($MN)
25 Global Sterile Injectable Market Outlook, By Musculoskeletal (2021-2030) ($MN)
26 Global Sterile Injectable Market Outlook, By Cancer (2021-2030) ($MN)
27 Global Sterile Injectable Market Outlook, By Infections (2021-2030) ($MN)
28 Global Sterile Injectable Market Outlook, By Diabetes (2021-2030) ($MN)
29 Global Sterile Injectable Market Outlook, By Central Nervous Systems (2021-2030) ($MN)
30 Global Sterile Injectable Market Outlook, By Other Applications (2021-2030) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We provide a free 15% customization on every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
Note: This customization is absolutely free until it falls under the 15% bracket. If your requirement exceeds this a feasibility check will be performed. Post that, a quote will be provided along with the timelines.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
