
Medical Device Market
Medical Device Market Forecasts to 2030 - Global Analysis By Product Type (Respiratory Devices, Cardiology Devices, Orthopedic Devices, Diagnostic Imaging Devices, Endoscopy Devices, Ophthalmology Devices, Wound Management Devices, Nephrology and Other Product Types), Application, End User and By Geography

|
Years Covered |
2021-2030 |
|
Estimated Year Value (2023) |
US $552.6 BN |
|
Projected Year Value (2030) |
US $941.0 BN |
|
CAGR (2023 - 2030) |
7.9% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
Asia Pacific |
According to Stratistics MRC, the Global Medical Device Market is accounted for $552.6 billion in 2023 and is expected to reach $941.0 billion by 2030 growing at a CAGR of 7.9% during the forecast period. An equipment, machine, implant, or in vitro reagent designed to be used in the identification, mitigation, monitoring, treatment, or prevention of illness or damage is referred to as a medical device. Medical devices come in a wide range of applications and levels of sophistication, but they are all vital to healthcare since they help with diagnosis and treatment of medical disorders. Manufacturers, retailers, and regulatory agencies all operate internationally in the medical device sector, which is a worldwide business.
According to 2022 statistics published by IDF, about 2.9 million people were living with diabetes in Canada in 2021. In addition, as per the same source, this number is projected to reach 3.2 million and 3.4 million by 2030 and 2045, respectively.
Market Dynamics:
Driver:
Rising prevalence of chronic diseases
The market for medical devices is significantly impacted by the growing incidence of chronic illnesses. Chronic illnesses frequently call for ongoing care and observation, including diabetes, respiratory conditions, cardiovascular diseases, and some forms of cancer. Additionally, remote patient monitoring technologies have advanced technologically due to the requirement for constant monitoring. Thus, healthcare providers can remotely monitor patients to medical gadgets that include connection characteristics like Bluetooth and internet access.
Restraint:
High cost
Expensive medical equipment may put a strain on healthcare finances, especially in systems that receive public funding. Moreover, the cost of purchasing and maintaining sophisticated technology can be prohibitive for hospitals and other healthcare institutions, which has an impact on their entire operating budget. Thus expensive medical equipment may be difficult for smaller healthcare institutions, such outpatient clinics or rural clinics, to afford and this makes it more difficult for them to offer a wide range of services, which might result in gaps in the delivery of healthcare thereby hampering the market growth
Opportunity:
Growing importance of home healthcare devices
There has been a surge in demand for home monitoring equipment, including glucometers, wearable fitness trackers, and blood pressure monitors and with the use of these gadgets, people may keep tabs on and take control of their health issues without constantly visiting medical institutions. Thus technology is always evolving due to the need for at-home medical equipment. To improve the entire patient experience, manufacturers are creating devices with intuitive user interfaces, connection features, and remote monitoring capabilities. Digital thermometers for use at home and telehealth platforms have become popular examples of devices that make remote consultations possible thereby propelling the growth of the market.
Threat:
Complex regulation requirements
Regulatory adjustments are expected to lead to higher costs for patient service offerings and the creation of new products. Among these advances related to secrecy of consumption are the General Data Protection Regulation (GDPR), which was put into effect by the European Union, and modifications to device approvals. The US FDA has asked for more clinical evidence to support the security and efficacy claims of medical equipment, such as endoscopic tools, which has resulted in a decreased extension of the time required to complete the marketing authorization process during the last ten years. The potential for missed revenues due to device release delays and higher costs as a result of onerous regulatory processes put pressure on investments in innovative devices, which affect the growth of the market for medical devices.
Covid-19 Impact
The COVID-19 pandemic affected the world market for medical equipment in a variety of ways. The unexpected increase in corona virus diagnoses around the globe has increased demand for medical equipment including respiration devices, ventilators, masks, personal protective equipment kits, and other items. Additionally, the use of digital health technologies like telemedicine and remote patient monitoring has increased dramatically during the COVID-19 epidemic. The market for medical equipment is expanding at a faster rate now. However, all non-essential surgical operations were put on hold during the peak days of COVID-19, giving preference to elective surgical treatments. This had a detrimental impact on the firms who make medical devices for necessary surgical procedures.
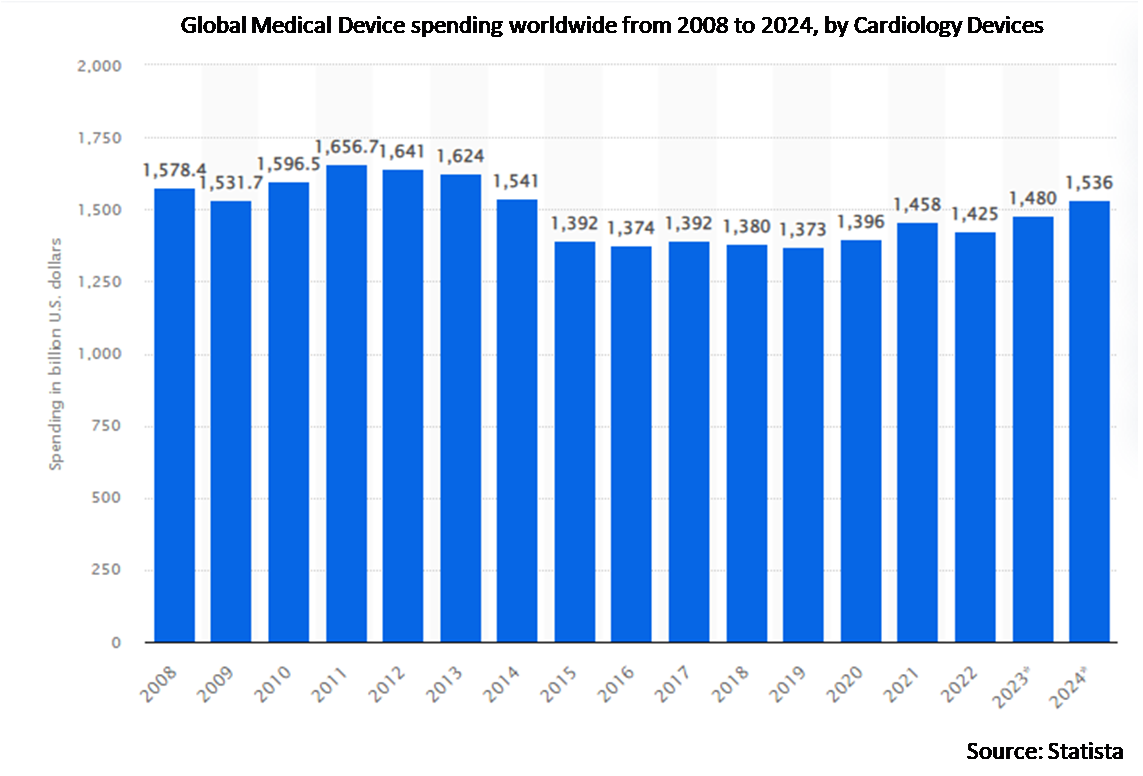
The cardiology devices segment is expected to be the largest during the forecast period
The cardiology devices segment is estimated to have a lucrative growth, due to many reasons, including the increasing incidence of cardiovascular illnesses, tactical moves by prominent industry players, and advancements in technology in cardiology devices. Furthermore, since more people with obesity, diabetes, hypertension, and high cholesterol live longer lives and are more prone to experience cardiac issues, these conditions are also driving up demand for cardiological medical equipment. Furthermore, an increasing number of patients are being managed with cardiology devices as a result of expanding business operations and technological advancements (such as the use of artificial intelligence in cardiac wearable devices), which has led to exponential therapeutical and monitoring outcomes are driving the growth of the market.
The diagnostic centers segment is expected to have the highest CAGR during the forecast period
The diagnostic centers segment is anticipated to witness the highest CAGR growth during the forecast period, as healthcare experts conduct tests and treatments in diagnostic centers in order to diagnose, track, and treat illnesses. Diagnostic facilities employ medical equipment for a variety of purposes. Diagnostic accuracy, speed, and efficiency are all improved in diagnostic centers by the incorporation of cutting-edge medical equipment. Additionally, it supports the early diagnosis, tracking, and customized treatment planning of diseases. Diagnostic centers' capacity to deliver high-quality healthcare services is continually shaped by the rapidly changing field of diagnostic technologies.
Region with largest share:
North America is projected to hold the largest market share during the forecast period owing to the Over the course of the forecast period, North America is anticipated to lead the medical devices market due to factors including the rising prevalence of chronic illnesses, high healthcare costs, and the presence of major companies. Furthermore, the medical device demand rises as a result of an increase in cardiopulmonary bypass procedures brought on by the rising prevalence of respiratory diseases like acute respiratory syndrome and chronic diseases like cardiovascular disease, coronary heart disease, and stroke in the general population. Thus the market is expanding as a result of businesses' increasing emphasis on creating cutting-edge goods and their adoption of diverse business tactics, such partnerships and acquisitions, to maintain their market positions.
Region with highest CAGR:
Asia Pacific is projected to have the highest CAGR over the forecast period, owing to factors such as an aging population, rising per capita income, and increased health consciousness among the populace, the Asia Pacific area grew at the quickest rate throughout the projected period. Moreover, the rising investments in the region's healthcare infrastructure are expected to support the growth of the medical device market. China and India are two of the major nations in the Asia Pacific area, ranking second and fifth respectively in the world market for revenue. In these nations, healthcare reforms and incentives provided by local governments mostly control the medical device industry.
Key players in the market
Some of the key players profiled in the Medical Device Market include Stryker, Medtronic, Fresenius SE & Co. KGaA, Abbott, Cardinal Health, BD, Koninklijke Philips N.V., General Electric Company, Siemens Healthineers AG, 3M Company, Boston Scientific Corporation, F. Hoffmann-La Roche Ltd, Smith & Nephew PLC, Braun Melsungen AG, Intuitive Surgical, Fujifilm, Canon Medical Systems, Alcon Laboratories, Edwards Lifesciences and Thermo Fisher
Key Developments:
In November 2023, Fresenius Kabi announced the launch of its tocilizumab biosimilar Tyenne® referencing RoActemra®** (tocilizumab) in the European Union. Tyenne® becomes the first tocilizumab biosimilar available in Europe for the treatment of several inflammatory and immune diseases.
In October 2023, Stryker one of the world’s leading medical technology companies, introduced Prime Connect the first smart, connected hospital stretcher designed to support fall prevention protocols from anywhere in a facility at the Emergency Nursing 2023 event in San Diego
In September 2023, Medtronic plc in healthcare technology, announced CE (Conformité Européenne) Mark approval for its new all-in-one, disposable Simplera™ continuous glucose monitor (CGM) featuring a simple, two-step insertion process.
Product Types Covered:
• Respiratory Devices
• Cardiology Devices
• Orthopedic Devices
• Diagnostic Imaging Devices
• Endoscopy Devices
• Ophthalmology Devices
• Wound Management Devices
• Nephrology
• Other Product Types
Applications Covered:
• Gastrointestinal Diseases
• Respiratory Disorders
• Cardiovascular Diseases
• Oral Health Conditions
• Neurological Disorders
• Common Orthopedic Disorders
• Other Applications
End Users Covered:
• Diagnostic Centers
• Research Laboratory
• Hospitals & Clinics
• Pharmaceutical Companies
• Other End Users
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2021, 2022, 2023, 2026, and 2030
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
o Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Product Analysis
3.7 Application Analysis
3.8 End User Analysis
3.9 Emerging Markets
3.10 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Medical Device Market, By Product Type
5.1 Introduction
5.2 Respiratory Devices
5.2.1 Non-Invasive Ventilatiors
5.2.2 Nasal Cannula
5.2.3 Oxygen Concentrator
5.2.4 Stethoscope
5.2.5 Continuous Positive Airway Pressure (CPAP) Device
5.2.6 Endotracheal Tubes
5.2.7 Inhalers
5.2.8 Humidifier
5.2.9 Portable/ Home oxygen concentrator
5.2.10 Other Respiratory Devices
5.3 Cardiology Devices
5.3.1 Implantable Cardioverter Defibrillators
5.3.2 Pacemakers
5.3.3 Biventricular Devices
5.3.4 Implantable Cardiac Loop Recorders
5.3.5 Sphygmomanometer
5.3.6 Cardiac Monitoring Devices
5.3.7 Pulmonary Artery Catheter
5.3.8 Atherectomy Catheters
5.3.9 Other Cardiology Devices
5.4 Orthopedic Devices
5.4.1 Orthopedic Implants
5.4.2 Screws
5.4.3 Braces
5.4.4 Bone plates
5.4.5 Suture Anchors
5.5 Diagnostic Imaging Devices
5.5.1 Computed Tomography (CT)
5.5.2 Magnetic Resonance Imaging (MRI)
5.5.3 Radiography (X-Rays)
5.5.4 Mammography
5.5.5 Ultrasound
5.5.6 Echocardiography
5.5.7 Fluoroscopy
5.5.8 Functional Magnetic Resonance Imaging
5.5.9 Scintigraphy
5.5.10 Other Diagnostic Imaging Devices
5.6 Endoscopy Devices
5.6.1 Endoscopy Cameras
5.6.2 Endoscopy Light Source
5.6.3 Endoscopy Video Processors
5.6.4 Insufflators
5.6.5 Gastroscopes
5.6.6 Insufflators
5.6.7 Colonoscopes
5.6.8 Capsule Endoscopes
5.6.9 Other Endoscopy Devices
5.7 Ophthalmology Devices
5.7.1 Tonometers
5.7.2 Retinoscopes
5.7.3 Keratoscope
5.7.4 Fixation Forceps
5.7.5 Fundus Camera
5.7.6 Optical Coherence Tomography
5.7.7 Other Ophthalmology Devices
5.8 Wound Management Devices
5.9 Nephrology
5.10 Other Product Types
6 Global Medical Device Market, By Application
6.1 Introduction
6.2 Gastrointestinal Diseases
6.3 Respiratory Disorders
6.4 Cardiovascular Diseases
6.5 Oral Health Conditions
6.6 Neurological Disorders
6.7 Common Orthopedic Disorders
6.8 Other Applications
7 Global Medical Device Market, By End User
7.1 Introduction
7.2 Diagnostic Centers
7.3 Research Laboratory
7.4 Hospitals & Clinics
7.5 Pharmaceutical Companies
7.6 Other End Users
8 Global Medical Device Market, By Geography
8.1 Introduction
8.2 North America
8.2.1 US
8.2.2 Canada
8.2.3 Mexico
8.3 Europe
8.3.1 Germany
8.3.2 UK
8.3.3 Italy
8.3.4 France
8.3.5 Spain
8.3.6 Rest of Europe
8.4 Asia Pacific
8.4.1 Japan
8.4.2 China
8.4.3 India
8.4.4 Australia
8.4.5 New Zealand
8.4.6 South Korea
8.4.7 Rest of Asia Pacific
8.5 South America
8.5.1 Argentina
8.5.2 Brazil
8.5.3 Chile
8.5.4 Rest of South America
8.6 Middle East & Africa
8.6.1 Saudi Arabia
8.6.2 UAE
8.6.3 Qatar
8.6.4 South Africa
8.6.5 Rest of Middle East & Africa
9 Key Developments
9.1 Agreements, Partnerships, Collaborations and Joint Ventures
9.2 Acquisitions & Mergers
9.3 New Product Launch
9.4 Expansions
9.5 Other Key Strategies
10 Company Profiling
10.1 Stryker
10.2 Medtronic
10.3 Fresenius SE & Co. KGaA
10.4 Abbott
10.5 Cardinal Health
10.6 BD
10.7 Koninklijke Philips N.V.
10.8 General Electric Company
10.9 Siemens Healthineers AG
10.10 3M Company
10.11 Boston Scientific Corporation
10.12 F. Hoffmann-La Roche Ltd
10.13 Smith & Nephew PLC
10.14 Braun Melsungen AG
10.15 Intuitive Surgical
10.16 Fujifilm
10.17 Canon Medical Systems
10.18 Alcon Laboratories
10.19 Edwards Lifesciences
10.20 Thermo Fisher
List of Tables
1 Global Medical Device Market Outlook, By Region (2021-2030) ($MN)
2 Global Medical Device Market Outlook, By Type (2021-2030) ($MN)
3 Global Medical Device Market Outlook, By Respiratory Devices (2021-2030) ($MN)
4 Global Medical Device Market Outlook, By Non-Invasive Ventilatiors (2021-2030) ($MN)
5 Global Medical Device Market Outlook, By Nasal Cannula (2021-2030) ($MN)
6 Global Medical Device Market Outlook, By Oxygen Concentrator (2021-2030) ($MN)
7 Global Medical Device Market Outlook, By Stethoscope (2021-2030) ($MN)
8 Global Medical Device Market Outlook, By Continuous Positive Airway Pressure (CPAP) Device (2021-2030) ($MN)
9 Global Medical Device Market Outlook, By Endotracheal Tubes (2021-2030) ($MN)
10 Global Medical Device Market Outlook, By Inhalers (2021-2030) ($MN)
11 Global Medical Device Market Outlook, By Humidifier (2021-2030) ($MN)
12 Global Medical Device Market Outlook, By Portable/ Home oxygen concentrator (2021-2030) ($MN)
13 Global Medical Device Market Outlook, By Other Respiratory Devices (2021-2030) ($MN)
14 Global Medical Device Market Outlook, By Cardiology Devices (2021-2030) ($MN)
15 Global Medical Device Market Outlook, By Implantable Cardioverter Defibrillators (2021-2030) ($MN)
16 Global Medical Device Market Outlook, By Pacemakers (2021-2030) ($MN)
17 Global Medical Device Market Outlook, By Biventricular Devices (2021-2030) ($MN)
18 Global Medical Device Market Outlook, By Implantable Cardiac Loop Recorders (2021-2030) ($MN)
19 Global Medical Device Market Outlook, By Sphygmomanometer (2021-2030) ($MN)
20 Global Medical Device Market Outlook, By Cardiac Monitoring Devices (2021-2030) ($MN)
21 Global Medical Device Market Outlook, By Pulmonary Artery Catheter (2021-2030) ($MN)
22 Global Medical Device Market Outlook, By Atherectomy Catheters (2021-2030) ($MN)
23 Global Medical Device Market Outlook, By Other Cardiology Devices (2021-2030) ($MN)
24 Global Medical Device Market Outlook, By Orthopedic Devices (2021-2030) ($MN)
25 Global Medical Device Market Outlook, By Orthopedic Implants (2021-2030) ($MN)
26 Global Medical Device Market Outlook, By Screws (2021-2030) ($MN)
27 Global Medical Device Market Outlook, By Braces (2021-2030) ($MN)
28 Global Medical Device Market Outlook, By Bone plates (2021-2030) ($MN)
29 Global Medical Device Market Outlook, By Suture Anchors (2021-2030) ($MN)
30 Global Medical Device Market Outlook, By Diagnostic Imaging Devices (2021-2030) ($MN)
31 Global Medical Device Market Outlook, By Computed Tomography (CT) (2021-2030) ($MN)
32 Global Medical Device Market Outlook, By Magnetic Resonance Imaging (MRI) (2021-2030) ($MN)
33 Global Medical Device Market Outlook, By Radiography (X-Rays) (2021-2030) ($MN)
34 Global Medical Device Market Outlook, By Mammography (2021-2030) ($MN)
35 Global Medical Device Market Outlook, By Ultrasound (2021-2030) ($MN)
36 Global Medical Device Market Outlook, By Echocardiography (2021-2030) ($MN)
37 Global Medical Device Market Outlook, By Fluoroscopy (2021-2030) ($MN)
38 Global Medical Device Market Outlook, By Functional Magnetic Resonance Imaging (2021-2030) ($MN)
39 Global Medical Device Market Outlook, By Scintigraphy (2021-2030) ($MN)
40 Global Medical Device Market Outlook, By Other Diagnostic Imaging Devices (2021-2030) ($MN)
41 Global Medical Device Market Outlook, By Endoscopy Devices (2021-2030) ($MN)
42 Global Medical Device Market Outlook, By Endoscopy Cameras (2021-2030) ($MN)
43 Global Medical Device Market Outlook, By Endoscopy Light Source (2021-2030) ($MN)
44 Global Medical Device Market Outlook, By Endoscopy Video Processors (2021-2030) ($MN)
45 Global Medical Device Market Outlook, By Insufflators (2021-2030) ($MN)
46 Global Medical Device Market Outlook, By Gastroscopes (2021-2030) ($MN)
47 Global Medical Device Market Outlook, By Insufflators (2021-2030) ($MN)
48 Global Medical Device Market Outlook, By Colonoscopes (2021-2030) ($MN)
49 Global Medical Device Market Outlook, By Capsule Endoscopes (2021-2030) ($MN)
50 Global Medical Device Market Outlook, By Other Endoscopy Devices (2021-2030) ($MN)
51 Global Medical Device Market Outlook, By Ophthalmology Devices (2021-2030) ($MN)
52 Global Medical Device Market Outlook, By Tonometers (2021-2030) ($MN)
53 Global Medical Device Market Outlook, By Retinoscopes (2021-2030) ($MN)
54 Global Medical Device Market Outlook, By Keratoscope (2021-2030) ($MN)
55 Global Medical Device Market Outlook, By Fixation Forceps (2021-2030) ($MN)
56 Global Medical Device Market Outlook, By Fundus Camera (2021-2030) ($MN)
57 Global Medical Device Market Outlook, By Optical Coherence Tomography (2021-2030) ($MN)
58 Global Medical Device Market Outlook, By Other Ophthalmology Devices (2021-2030) ($MN)
59 Global Medical Device Market Outlook, By Wound Management Devices (2021-2030) ($MN)
60 Global Medical Device Market Outlook, By Nephrology (2021-2030) ($MN)
61 Global Medical Device Market Outlook, By Other Types (2021-2030) ($MN)
62 Global Medical Device Market Outlook, By Application (2021-2030) ($MN)
63 Global Medical Device Market Outlook, By Gastrointestinal Diseases (2021-2030) ($MN)
64 Global Medical Device Market Outlook, By Respiratory Disorders (2021-2030) ($MN)
65 Global Medical Device Market Outlook, By Cardiovascular Diseases (2021-2030) ($MN)
66 Global Medical Device Market Outlook, By Oral Health Conditions (2021-2030) ($MN)
67 Global Medical Device Market Outlook, By Neurological Disorders (2021-2030) ($MN)
68 Global Medical Device Market Outlook, By Common Orthopedic Disorders (2021-2030) ($MN)
69 Global Medical Device Market Outlook, By Other Applications (2021-2030) ($MN)
70 Global Medical Device Market Outlook, By End User (2021-2030) ($MN)
71 Global Medical Device Market Outlook, By Diagnostic Centers (2021-2030) ($MN)
72 Global Medical Device Market Outlook, By Research Laboratory (2021-2030) ($MN)
73 Global Medical Device Market Outlook, By Hospitals & Clinics (2021-2030) ($MN)
74 Global Medical Device Market Outlook, By Pharmaceutical Companies (2021-2030) ($MN)
75 Global Medical Device Market Outlook, By Other End Users (2021-2030) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We offer complimentary customization of up to 15% with every purchase. To share your customization requirements, feel free to email us at info@strategymrc.com or call us on +1-301-202-5929. .
Please Note: Customization within the 15% threshold is entirely free of charge. If your request exceeds this limit, we will conduct a feasibility assessment. Following that, a detailed quote and timeline will be provided.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
