
Real World Evidence Market
Real-World Evidence Market Forecasts to 2030 - Global Analysis By Component (Services and Data Sets), By Application, By End User and By Geography

|
Years Covered |
2021-2030 |
|
Estimated Year Value (2023) |
US $1.7 BN |
|
Projected Year Value (2030) |
US $4.5 BN |
|
CAGR (2023 - 2030) |
14.8% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
Asia Pacific |
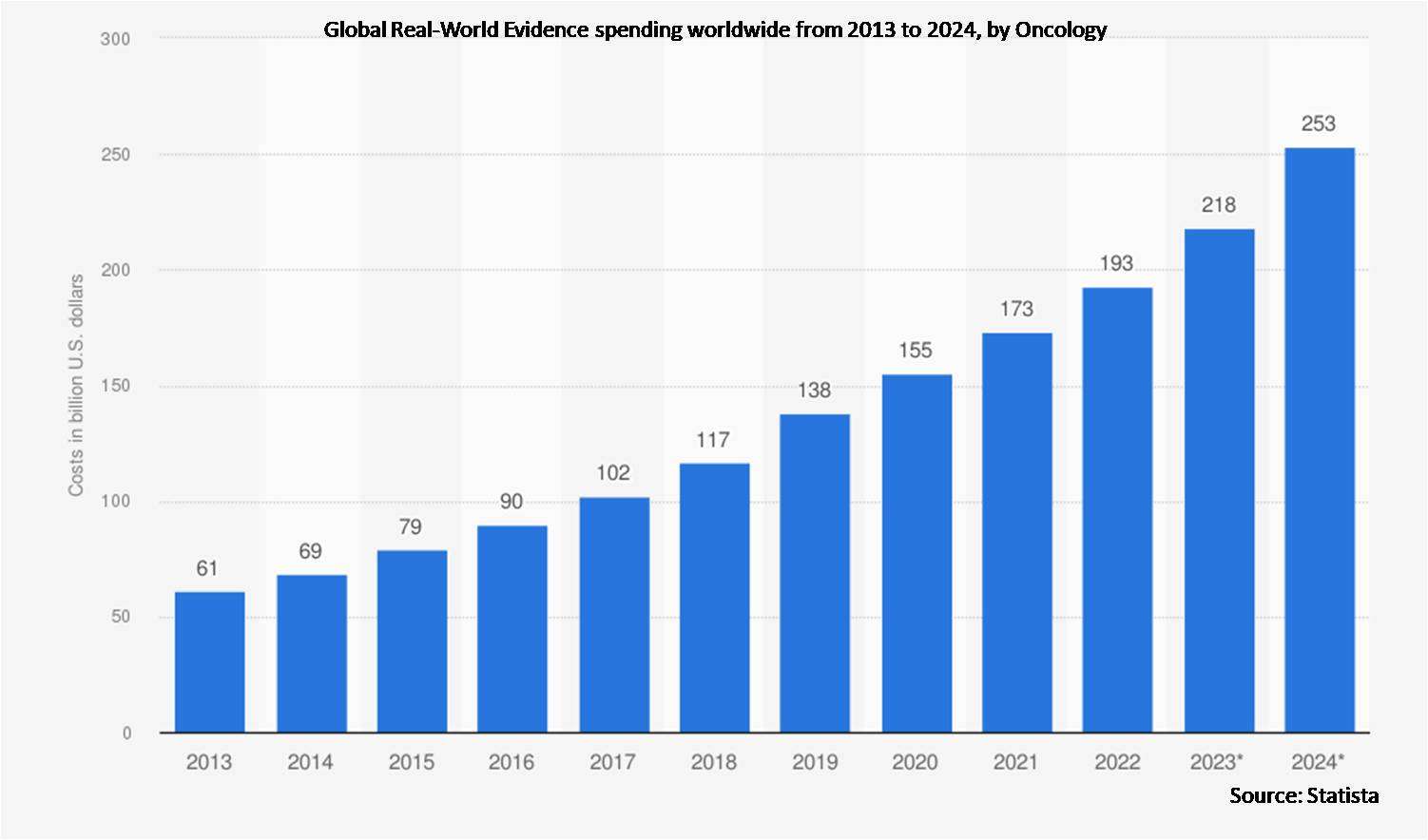
According to Stratistics MRC, the Global Real-World Evidence Market is accounted for $1.7 billion in 2023 and is expected to reach $4.5 billion by 2030 growing at a CAGR of 14.8% during the forecast period. Real-world evidence (RWE) is data collected outside traditional clinical trials, providing insights into a treatment's effectiveness in real-world settings. Its uses include informing healthcare decisions, understanding treatment outcomes, and assessing long-term patient outcomes.
According to Clinicaltrials.gov, as of February 2022, there are around 87,366 cancer clinical trials across different phases of development.
Market Dynamics:
Driver:
Cost-efficiency and time savings
Compared to traditional clinical trials, utilizing real-world data for research and decision-making significantly reduces the time and financial investments required. RWE allows for continuous monitoring of treatment outcomes in diverse populations, providing timely insights at a fraction of the cost. This efficiency is particularly appealing to pharmaceutical companies, regulators, and healthcare stakeholders, fostering a shift toward evidence generation that is not only more economical but also aligns with the increasing demand for agile and adaptive approaches in the evolving healthcare landscape.
Restraint:
Data quality and standardization
Variability in data collection methods, formats, and quality across diverse healthcare systems can result in inconsistencies and limitations in the reliability of real-world data. Incomplete or non-standardized information hinders the robustness of RWE, impacting its credibility for decision-making. Efforts to establish standardized data collection practices, improve data quality through validation processes, and enhance interoperability among different healthcare systems are imperative.
Opportunity:
Expanded focus on patient-centric healthcare
Healthcare systems increasingly prioritize patient outcomes and experiences, the demand for real-world data to understand treatment effectiveness, safety, and patient preferences grows. RWE offers a valuable tool to capture insights directly from patients' everyday experiences, aligning with the broader shift towards personalized and patient-centered care. The integration of patient-reported outcomes and real-world evidence enhances the holistic understanding of treatments, fostering more patient-centric decision-making and facilitating the development of therapies that better meet the needs and expectations of diverse patient populations.
Threat:
Privacy and security concerns
The utilization of sensitive patient data raises ethical and regulatory challenges, with the potential for breaches compromising patient confidentiality. Striking a delicate balance between extracting valuable insights from real-world data and safeguarding patient privacy becomes crucial. Stringent data protection regulations, like GDPR and HIPAA, impose strict requirements, adding complexity to RWE initiatives. The threat of data breaches not only jeopardizes patient trust but also raises legal and compliance issues for stakeholders.
Covid-19 Impact:
The Covid-19 pandemic has accelerated the importance of Real-World Evidence (RWE) in healthcare decision-making. The urgent need for rapid insights into treatment effectiveness and outcomes has underscored the value of leveraging real-world data. The pandemic has prompted increased collaboration between stakeholders, including regulatory bodies, to utilize RWE for evaluating the impact of interventions. While the crisis has highlighted the potential of RWE, it has also revealed challenges such as data quality issues and the need for standardized methodologies, emphasizing the ongoing efforts required to fully harness the potential of real-world evidence in a public health crisis.
The drug development and approvals segment is expected to be the largest during the forecast period
Drug development and approvals in the market is expected to hold a significant share. Pharmaceutical companies leverage RWE to complement traditional clinical trial data, offering a more comprehensive understanding of a drug's effectiveness and safety across diverse patient populations. Regulatory bodies increasingly recognize the significance of RWE in making informed decisions during the drug approval process, enabling a more efficient and patient-centered approach to drug development, regulatory submissions, and post-market surveillance. The integration of RWE enhances the evidence base, contributing to more informed healthcare decisions and improved patient outcomes.
The pharmaceutical & medical device companies segment is expected to have the highest CAGR during the forecast period
During the anticipated period, the pharmaceutical & medical device companies segment is expected to increase at the most effective rate. RWE enables these companies to assess the effectiveness and safety of treatments in diverse patient populations, supporting regulatory submissions and market access. It facilitates a more comprehensive understanding of product performance beyond controlled clinical trials, influencing decision-making throughout the product lifecycle. The integration of RWE empowers pharmaceutical and medical device companies to make informed strategic decisions, navigate regulatory landscapes, and demonstrate the real-world impact of their innovations in healthcare.
Region with largest share:
During the forecast period, it is expected that the North American market will hold a majority of the market share. The region's advanced electronic health records (EHR) systems and widespread adoption of health informatics contribute to a rich source of real-world data. Regulatory support for incorporating RWE into decision-making processes, coupled with a strong emphasis on healthcare innovation, positions North America at the forefront of leveraging real-world evidence to inform clinical practices, drug development, and health policy decisions.
Region with highest CAGR:
Asia Pacific is projected to have the highest CAGR over the forecast period. Increasing healthcare digitization, rising demand for evidence-based decision-making, and expanding healthcare infrastructures contribute to the region's adoption of RWE. The diverse patient populations across countries present an opportunity to generate valuable insights. However, challenges include varying regulatory landscapes and data privacy concerns. As the region continues to prioritize healthcare advancements, collaborations, and technology integration, the utilization of real-world evidence is poised to play a pivotal role in shaping healthcare strategies and policies in Asia Pacific.
Key players in the market
Some of the key players in Real-World Evidence market include Cegedim Health Data, PerkinElmer, Inc., IBM, Syneos Health, Icon Plc., PPD, Inc., IQVIA, Medpace, Parexel International Corporation and Oracle.
Key Developments:
In August 2022, Trinity Life Sciences (US), announced a partnership with Bain & Company (US). The partnership is intended to pair the extensive experience of Bain & Company (US) in the Life Sciences value chain with the evidence-based solutions of Trinity Life Sciences (US).
In July 2022, TriNetX LLC announced the collaboration with Pediatric Medical Centers (US), to help Pediatric Medical Centers including Children’s National Hospital (US), WVU Medicine Children’s (US), and John Hopkins All Children Hospitals (US), to improvise the investor-initiated research utilizing the pediatric real-world data.
In June 2022, IQVIA partnered with Argenex SE for accelerating the clinical development of new indications for VYVGART. Furthermore, it will leverage its scientific advice and technology in supporting clinical development by Argenx.
Components Covered:
• Services
• Data Sets
Applications Covered:
• Drug Development And Approvals
• Post-Market Surveillance
• Market Access Reimbursement/Coverage Decision Making
• Clinical And Regulatory Decision-Making
• Other Applications
End Users Covered:
• Pharmaceutical & Medical Device Companies
• Healthcare Providers
• Healthcare Payers
• Other End Users
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2021, 2022, 2023, 2026, and 2030
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Application Analysis
3.7 End User Analysis
3.8 Emerging Markets
3.9 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Real-World Evidence Market, By Component
5.1 Introduction
5.2 Services
5.3 Data Sets
5.3.1 Disparate Data Sets
5.3.1.1 Clinical Settings Data
5.3.1.2 Claims Data
5.3.1.3 Pharmacy Data
5.3.1.4 Patient-Powered Data
5.3.1.5 Registry-Based Data Sets
5.3.2 Integrated Data Sets
6 Global Real-World Evidence Market, By Application
6.1 Introduction
6.2 Drug Development And Approvals
6.2.1 Oncology
6.2.2 Neurology
6.2.3 Immunology
6.2.4 Cardiovascular Disease
6.2.5 Other Therapeutic Areas
6.2.6 Medical Device Development And Approvals
6.3 Post-Market Surveillance
6.4 Market Access Reimbursement/Coverage Decision Making
6.5 Clinical And Regulatory Decision-Making
6.6 Other Applications
7 Global Real-World Evidence Market, By End User
7.1 Introduction
7.2 Pharmaceutical & Medical Device Companies
7.3 Healthcare Providers
7.4 Healthcare Payers
7.5 Other End Users
8 Global Real-World Evidence Market, By Geography
8.1 Introduction
8.2 North America
8.2.1 US
8.2.2 Canada
8.2.3 Mexico
8.3 Europe
8.3.1 Germany
8.3.2 UK
8.3.3 Italy
8.3.4 France
8.3.5 Spain
8.3.6 Rest of Europe
8.4 Asia Pacific
8.4.1 Japan
8.4.2 China
8.4.3 India
8.4.4 Australia
8.4.5 New Zealand
8.4.6 South Korea
8.4.7 Rest of Asia Pacific
8.5 South America
8.5.1 Argentina
8.5.2 Brazil
8.5.3 Chile
8.5.4 Rest of South America
8.6 Middle East & Africa
8.6.1 Saudi Arabia
8.6.2 UAE
8.6.3 Qatar
8.6.4 South Africa
8.6.5 Rest of Middle East & Africa
9 Key Developments
9.1 Agreements, Partnerships, Collaborations and Joint Ventures
9.2 Acquisitions & Mergers
9.3 New Product Launch
9.4 Expansions
9.5 Other Key Strategies
10 Company Profiling
10.1 Cegedim Health Data
10.2 PerkinElmer, Inc.
10.3 IBM
10.4 Syneos Health
10.5 Icon Plc.
10.6 PPD, Inc.
10.7 IQVIA
10.8 Medpace
10.9 Parexel International Corporation
10.10 Oracle
List of Tables
1 Global Real-World Evidence Market Outlook, By Region (2021-2030) ($MN)
2 Global Real-World Evidence Market Outlook, By Component (2021-2030) ($MN)
3 Global Real-World Evidence Market Outlook, By Services (2021-2030) ($MN)
4 Global Real-World Evidence Market Outlook, By Data Sets (2021-2030) ($MN)
5 Global Real-World Evidence Market Outlook, By Disparate Data Sets (2021-2030) ($MN)
6 Global Real-World Evidence Market Outlook, By Clinical Settings Data (2021-2030) ($MN)
7 Global Real-World Evidence Market Outlook, By Claims Data (2021-2030) ($MN)
8 Global Real-World Evidence Market Outlook, By Pharmacy Data (2021-2030) ($MN)
9 Global Real-World Evidence Market Outlook, By Patient-Powered Data (2021-2030) ($MN)
10 Global Real-World Evidence Market Outlook, By Registry-Based Data Sets (2021-2030) ($MN)
11 Global Real-World Evidence Market Outlook, By Integrated Data Sets (2021-2030) ($MN)
12 Global Real-World Evidence Market Outlook, By Application (2021-2030) ($MN)
13 Global Real-World Evidence Market Outlook, By Drug Development And Approvals (2021-2030) ($MN)
14 Global Real-World Evidence Market Outlook, By Oncology (2021-2030) ($MN)
15 Global Real-World Evidence Market Outlook, By Neurology (2021-2030) ($MN)
16 Global Real-World Evidence Market Outlook, By Immunology (2021-2030) ($MN)
17 Global Real-World Evidence Market Outlook, By Cardiovascular Disease (2021-2030) ($MN)
18 Global Real-World Evidence Market Outlook, By Other Therapeutic Areas (2021-2030) ($MN)
19 Global Real-World Evidence Market Outlook, By Medical Device Development And Approvals (2021-2030) ($MN)
20 Global Real-World Evidence Market Outlook, By Post-Market Surveillance (2021-2030) ($MN)
21 Global Real-World Evidence Market Outlook, By Market Access Reimbursement/Coverage Decision Making (2021-2030) ($MN)
22 Global Real-World Evidence Market Outlook, By Clinical And Regulatory Decision-Making (2021-2030) ($MN)
23 Global Real-World Evidence Market Outlook, By Other Applications (2021-2030) ($MN)
24 Global Real-World Evidence Market Outlook, By End User (2021-2030) ($MN)
25 Global Real-World Evidence Market Outlook, By Pharmaceutical & Medical Device Companies (2021-2030) ($MN)
26 Global Real-World Evidence Market Outlook, By Healthcare Providers (2021-2030) ($MN)
27 Global Real-World Evidence Market Outlook, By Healthcare Payers (2021-2030) ($MN)
28 Global Real-World Evidence Market Outlook, By Other End Users (2021-2030) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We offer complimentary customization of up to 15% with every purchase. To share your customization requirements, feel free to email us at info@strategymrc.com or call us on +1-301-202-5929. .
Please Note: Customization within the 15% threshold is entirely free of charge. If your request exceeds this limit, we will conduct a feasibility assessment. Following that, a detailed quote and timeline will be provided.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
