
Bionic Devices Market
Bionic Devices Market Forecasts to 2028 – Global Analysis By Product (Ear Bionics, Cardiac Bionics), Fixation (Externally Worn, Implantable), End User, and By Geography

|
Years Covered |
2020-2028 |
|
Estimated Year Value (2021) |
US $5.08 BN |
|
Projected Year Value (2028) |
US $9.96 BN |
|
CAGR (2021 - 2028) |
10.1% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia Pacific |
According to Stratistics MRC, the Global Bionic Devices Market is accounted for $5.08 billion in 2021 and is expected to reach $9.96 billion by 2028 growing at a CAGR of 10.1% during the forecast period. Bionics are artificial devices that can be implanted or integrated into humans to either replace a natural organ or provide support to restore a specific function, or a group of related functions, helping the patient to return to his normal life.
The increasing number of transplants, coupled with the rising number of people waiting for donors and large global population with heart diseases are providing ample opportunities for the artificial organ and bionics market. According to the Human Resource and Services Administration (HRSA), in 2021, 41,354 organ transplants were performed in the United States, an increase of 5.9 percent over 2020 and the first time the annual total exceeded 40,000, according to preliminary data from the Organ Procurement and Transplantation Network.
Market Dynamics:
Driver:
Growing incidence of chronic conditions
The rising prevalence of chronic conditions that promote amputations is likely to expand the requirement for bionic devices. Several chronic conditions are liable for amputation. Patients suffering from diabetes are exposed to multiple times higher risk of undergoing amputation and are responsible for the significant economic strain on healthcare systems. Trauma is another chronic condition that is related to the rising incidence of amputation for the lower extremity. The rate of these chronic conditions is directly relative to the rise in the number of amputations. Because of the high prevalence of chronic conditions, the number of amputations is constantly surging.
Restraint:
High cost
Bionic devices are costly because of the highly expensive devices and techniques. Recent advances in robotic prosthetic hands have been a little short of extraordinary, but have remained out of the price range of many who need them. This is due to many factors, but the primary one is that different components of bionic devices are traditionally manufactured in different facilities, by different companies.
Opportunity:
Technological Advancements
The field of medical bionics has witnessed several technological advancements in recent years. The cochlear implants available in the current situation have speech recognition in difficult listening circumstances, which help to separate speech in noisy and reverberant situations, grasp sound from television or telephone, and identify a sound from distance. Furthermore, hearing aids feature acoustic scene classifiers, which assist hearing aids to classify and recognize any sound based on background noise, speech in quiet or in noise, music, wind, and several other parameters.
Threat:
Limitation of metal electrodes
Bionic devices regularly utilize metal electrodes to stimulate the nerve cells. While these electrodes have been sufficient for the past few decades and in devices that are implanted for only a few years, these metal electrodes are currently turning into a limitation. As the miniaturization of electronics continues to allow us to reduce the size of implants and offer patients devices with more functionality, there is a need to create smaller, more densely packed electrodes. At these sub-micron sizes, metal electrodes cannot be securely utilized for multiple decades, as is currently expected by cochlear implant patients who receive devices as children.
The implantable segment is expected to be the largest during the forecast period
The implantable segment is estimated to have a lucrative growth owing to the increasing cases of severe road accidents and physical injuries, rapid adoption of advanced bionic devices, and growing demand for implantable bio-implants because of various advantages over external bionics, and rising healthcare expenditure.
The ear bionics segment is expected to have the highest CAGR during the forecast period
The ear bionics segment is anticipated to witness the fastest CAGR growth during the forecast period due to the rapidly growing geriatric population, rising volumes of ear surgeries, increasing incidences of hearing disorders, and growing use of advanced cochlear implants, bone-anchored hearing systems, and auditory brainstem implants.
Region with highest share:
North America is projected to hold the largest market share during the forecast period due to the growing geriatric population, increasing occurrences of road accidents and irreversible physical injuries, and the emergence of highly advanced bionic technologies. Market growth is further boosted by factors such as a rise in healthcare expenditure, growing per capita income, and the presence of avant-garde healthcare infrastructure in the region.
Region with highest CAGR:
Asia Pacific is projected to have the highest CAGR over the forecast period owing to emerging economies like Japan, China, and India. These countries have a large population base with a high incidence of chronic diseases. An increase in the number of chronic diseases that lead to organ failure is expected to boost artificial organ and bionics device growth.
Key players in the market
Some of the key players profiled in the Bionic Devices Market include Touch bionics (Ossur Corporate), Syncardia, Medtronic, Scheck & Siress, ReWalk Robotics, Open Bionics, MED-EL Medical Electronics, Ekso Bionics, Demant A/S, Cochlear Ltd, Boston Scientific, Abbott, Sonova and Widex.
Key Developments:
In October 2020, Medtronic Private Limited launched the Azure pacemaker with BlueSync technology that can communicate directly with patients’ smartphones and tablets. It offers improved longevity, so patients potentially need fewer device replacements and allows patients to have MRI scans in either 1.5 or 3 Tesla (T) machines.
In March 2020, Widex launched the Moment hearing device. The Widex Moment has the ‘classic’ Widex signal pathway, which is paired with a second ultra-fast signal pathway that adds extra gearing to the platform.
Products Covered:
• Vision Bionics
• Ear Bionics
• Orthopedic Bionic
• Cardiac Bionics
• Neural Bionics
Fixations Covered:
• Wearable/ Externally Worn
• Implantable
Materials Covered:
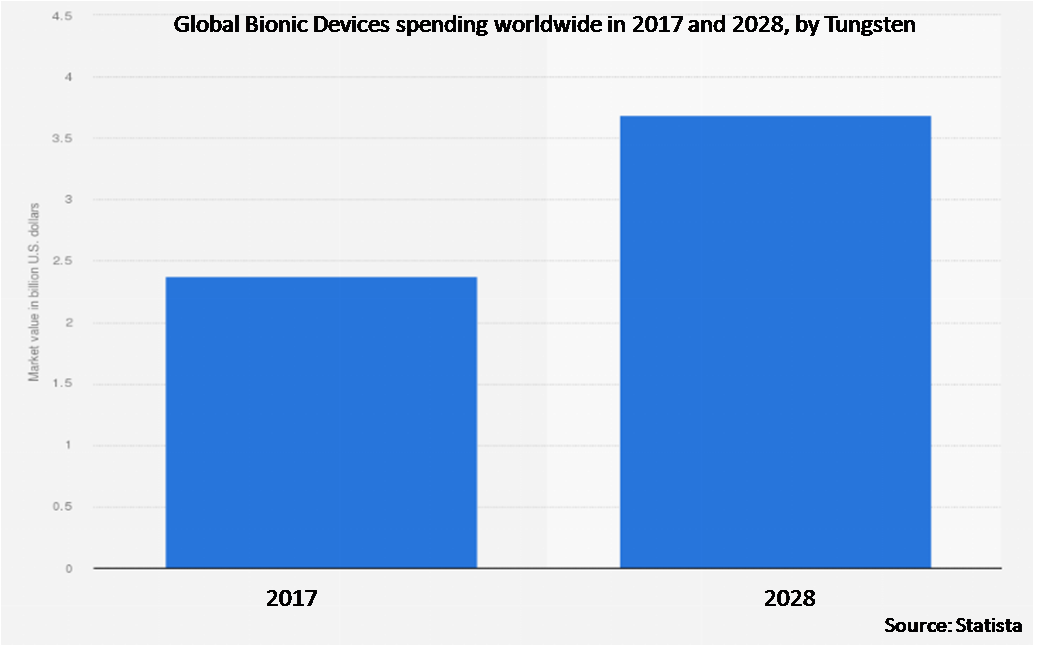
• Tungsten
• Stainless Steel
• Platinum-Iridium
• Biologics
• Silicon
Technologies Covered:
• Manual
• Robotic
End Users Covered:
• Research & Academic Institutes
• Hospitals & Clinics
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2020, 2021, 2022, 2025, and 2028
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
o Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Product Analysis
3.7 End User Analysis
3.8 Emerging Markets
3.9 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Bionic Devices Market, By Product
5.1 Introduction
5.2 Vision Bionics
5.3 Ear Bionics
5.3.1 Cochlear Implant
5.3.2 Bone-Anchored Hearing Aid
5.4 Orthopedic Bionic
5.4.1 Bionic Hand
5.4.2 Bionic Leg & Socket
5.4.3 Exoskeletons
5.5 Cardiac Bionics
5.5.1 Heart Valves
5.5.2 Ventricular-Assisted Device
5.5.3 Pacemaker
5.6 Neural Bionics
5.6.1 Spinal Cord Stimulator
5.6.2 Nerve Stimulation
5.6.3 Deep Brain Stimulator
6 Global Bionic Devices Market, By Fixation
6.1 Introduction
6.2 Wearable/ Externally Worn
6.3 Implantable
7 Global Bionic Devices Market, By Material
7.1 Introduction
7.2 Tungsten
7.3 Stainless Steel
7.4 Platinum-Iridium
7.5 Biologics
7.6 Silicon
8 Global Bionic Devices Market, By Technology
8.1 Introduction
8.2 Manual
8.3 Robotic
9 Global Bionic Devices Market, By End User
9.1 Introduction
9.2 Research & Academic Institutes
9.3 Hospitals & Clinics
10 Global Bionic Devices Market, By Geography
10.1 Introduction
10.2 North America
10.2.1 US
10.2.2 Canada
10.2.3 Mexico
10.3 Europe
10.3.1 Germany
10.3.2 UK
10.3.3 Italy
10.3.4 France
10.3.5 Spain
10.3.6 Rest of Europe
10.4 Asia Pacific
10.4.1 Japan
10.4.2 China
10.4.3 India
10.4.4 Australia
10.4.5 New Zealand
10.4.6 South Korea
10.4.7 Rest of Asia Pacific
10.5 South America
10.5.1 Argentina
10.5.2 Brazil
10.5.3 Chile
10.5.4 Rest of South America
10.6 Middle East & Africa
10.6.1 Saudi Arabia
10.6.2 UAE
10.6.3 Qatar
10.6.4 South Africa
10.6.5 Rest of Middle East & Africa
11 Key Developments
11.1 Agreements, Partnerships, Collaborations and Joint Ventures
11.2 Acquisitions & Mergers
11.3 New Product Launch
11.4 Expansions
11.5 Other Key Strategies
12 Company Profiling
12.1 Touch bionics (Ossur Corporate)
12.2 Syncardia
12.3 Medtronic
12.4 Scheck & Siress
12.5 ReWalk Robotics
12.6 Open Bionics
12.7 MED-EL Medical Electronics
12.8 Ekso Bionics
12.9 Demant A/S
12.10 Cochlear Ltd
12.11 Boston Scientific
12.12 Abbott
12.13 Sonova
12.14 Widex
List of Tables
1 Global Bionic Devices Market Outlook, By Region (2020-2028) ($MN)
2 Global Bionic Devices Market Outlook, By Product (2020-2028) ($MN)
3 Global Bionic Devices Market Outlook, By Vision Bionics (2020-2028) ($MN)
4 Global Bionic Devices Market Outlook, By Ear Bionics (2020-2028) ($MN)
5 Global Bionic Devices Market Outlook, By Cochlear Implant (2020-2028) ($MN)
6 Global Bionic Devices Market Outlook, By Bone-Anchored Hearing Aid (2020-2028) ($MN)
7 Global Bionic Devices Market Outlook, By Orthopedic Bionic (2020-2028) ($MN)
8 Global Bionic Devices Market Outlook, By Bionic Hand (2020-2028) ($MN)
9 Global Bionic Devices Market Outlook, By Bionic Leg & Socket (2020-2028) ($MN)
10 Global Bionic Devices Market Outlook, By Exoskeletons (2020-2028) ($MN)
11 Global Bionic Devices Market Outlook, By Cardiac Bionics (2020-2028) ($MN)
12 Global Bionic Devices Market Outlook, By Heart Valves (2020-2028) ($MN)
13 Global Bionic Devices Market Outlook, By Ventricular-Assisted Device (2020-2028) ($MN)
14 Global Bionic Devices Market Outlook, By Pacemaker (2020-2028) ($MN)
15 Global Bionic Devices Market Outlook, By Neural Bionics (2020-2028) ($MN)
16 Global Bionic Devices Market Outlook, By Spinal Cord Stimulator (2020-2028) ($MN)
17 Global Bionic Devices Market Outlook, By Nerve Stimulation (2020-2028) ($MN)
18 Global Bionic Devices Market Outlook, By Deep Brain Stimulator (2020-2028) ($MN)
19 Global Bionic Devices Market Outlook, By Fixation (2020-2028) ($MN)
20 Global Bionic Devices Market Outlook, By Wearable/ Externally Worn (2020-2028) ($MN)
21 Global Bionic Devices Market Outlook, By Implantable (2020-2028) ($MN)
22 Global Bionic Devices Market Outlook, By Material (2020-2028) ($MN)
23 Global Bionic Devices Market Outlook, By Tungsten (2020-2028) ($MN)
24 Global Bionic Devices Market Outlook, By Stainless Steel (2020-2028) ($MN)
25 Global Bionic Devices Market Outlook, By Platinum-Iridium (2020-2028) ($MN)
26 Global Bionic Devices Market Outlook, By Biologics (2020-2028) ($MN)
27 Global Bionic Devices Market Outlook, By Silicon (2020-2028) ($MN)
28 Global Bionic Devices Market Outlook, By Technology (2020-2028) ($MN)
29 Global Bionic Devices Market Outlook, By Manual (2020-2028) ($MN)
30 Global Bionic Devices Market Outlook, By Robotic (2020-2028) ($MN)
31 Global Bionic Devices Market Outlook, By End User (2020-2028) ($MN)
32 Global Bionic Devices Market Outlook, By Research & Academic Institutes (2020-2028) ($MN)
33 Global Bionic Devices Market Outlook, By Hospitals & Clinics (2020-2028) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We offer complimentary customization of up to 15% with every purchase. To share your customization requirements, feel free to email us at info@strategymrc.com or call us on +1-301-202-5929. .
Please Note: Customization within the 15% threshold is entirely free of charge. If your request exceeds this limit, we will conduct a feasibility assessment. Following that, a detailed quote and timeline will be provided.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
