
Laboratory Developed Testing Market
Laboratory-Developed Testing Market Forecasts to 2028 – Global Analysis By Test Type (Microbiology, Immunology, Haematology, Molecular Diagnostics), Product (R&D, Mass Spectrometry, Microarrays, PCR, NGS), Application (Genetics, Oncology, Infectious Diseases, Neurology), End User (Specialty Diagnostics Centers, Academic Institutes, Hospital Laboratories), and By Geography

|
Years Covered |
2020-2028 |
|
Estimated Year Value (2022) |
US $4.88 BN |
|
Projected Year Value (2028) |
US $8.45 BN |
|
CAGR (2022 - 2028) |
9.6% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
Asia Pacific |
According to Stratistics MRC, the Global Laboratory-Developed Testing Market is accounted for $4.88 billion in 2022 and is expected to reach $8.45 billion by 2028 growing at a CAGR of 9.6% during the forecast period. Laboratory developed test is a type of in vitro diagnostic test that is planned and utilized within a single laboratory. These tests can be used to gauge or discriminate a wide variety of analytes materials such as proteins, chemical compounds like cholesterol or glucose, or DNA, from a specimen received from human anatomy. The development of automated in vitro diagnostics (IVD) techniques for labs and dispensaries to deliver exact and error-free analysis is expected to fuel the increment. Laboratory developed tests are beneficial for the detection and monitoring of oncology, genetics, infections, and autoimmune diseases. These tests are the best option for the population curious about their genetic makeup concerning the presence of any disease-related markers. This testing offers numerous advantages such as improved quality of life, monitoring pre-existing conditions, and maintaining one’s lifestyle accordingly, which makes them preferable over hospitalization for daily checkups or the treatment of disease occurrence.
According to the American Heart Association, in 2017, 868,662 people died due to various forms of cardiovascular diseases in the U.S. Moreover, in 2018, coronary heart disease (42.1%) was one of the leading causes of death along with high blood pressure (11%), stroke (17%), arteries diseases (2.9%), and heart failure (9.6%). Therefore, for detection and treatment of these cardiovascular diseases, in-vitro diagnostics methods are highly preferred by the doctors. In addition, As per American Cancer Society, in 2020, about 1.8 million new cancer cases were diagnosed, and approximately 606,520 cancer deaths occurred in the United States. Moreover, people with chronic conditions are the most frequent users of healthcare facilities in the United States.
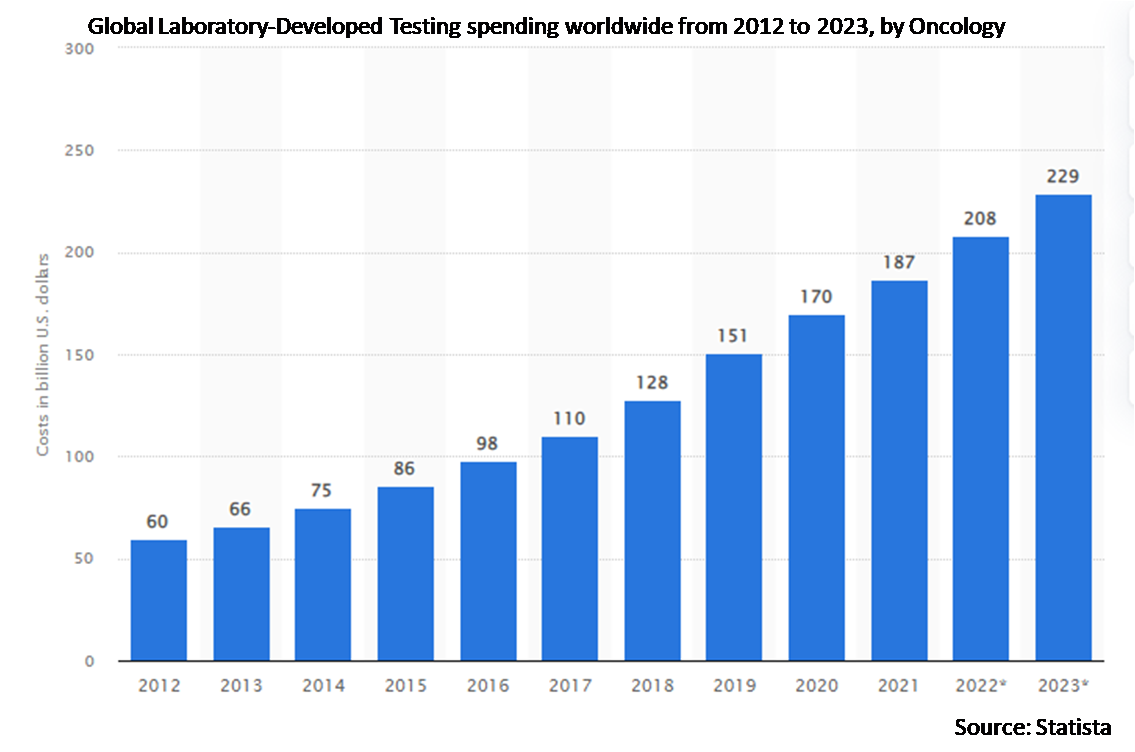
Market Dynamics:
Driver:
Increasing product offerings related to genetic health risks
Manufacturers of laboratory-developed tests across the world will have ample of lucrative prospects in the future. They have begun to recognize the value of R&D spending to provide a diversified product line. For instance, 23andMe and Virgin Group's VG Acquisition Corp. merged to become a publicly-traded company that will transform personalized healthcare and therapeutic advancement using human genetics. Many companies are taking extensive measures to make their product offerings more versatile through innovations, such as providing data related to genetic health risks, ancestry, and traits.
Restraint:
Stringent regulations
The in vitro diagnostic medical devices regulation (IVDR) will produce results in May 2022. This rule has a large impact on both the clinical laboratories and manufacturers of in vitro diagnostic medical devices. For clinical laboratories, the IVDR poses limitations on the exploit of laboratory developed tests (LDTs). To give a uniform interpretation of the IVDR for contemporaries in clinical practice, the IVDR task force was created by the scientific societies of laboratory specialties. A guidance document with explanations and interpretations of pertinent passages of the IVDR was drafted to assist laboratories prepare for the impact of this new legislation. With new regulations, a massive amount of work will be required to transition from the IVDD to the IVDR. IVDR is more intricate and detailed and will need more resources to uphold and implement for IVD manufacturers.
Opportunity:
Continuous research on personalized medicines provides growth opportunities to laboratory developed test market players
LDTs play a crucial function in the expansion of personalized medicines that are probable to show as hopeful means of handling diseases during distant eluded effectual treatments or cures. According to the Personalized Medicine Coalition, personalized medicines accounted for only 5% of the new FDA-approved molecular entities in 2005; though, in 2016, this numeral rose to more than 25%. Moreover, 42% of all compounds and 73% of oncology compounds in the pipeline have the potential to serve as personalized medicines. Biopharmaceutical companies have nearly doubled their R&D investments in personalized drugs in the last five years, and they are additionally anticipated to augment their investments by 33% in the next five years. Biopharmaceutical researchers also forecast a 69% increase in the expansion of personalized medicines in the next five years. Laboratory tests are used to diagnose disease and foresee and monitor drug response as well as to get informatics data required for complex predictive algorithms. Personalized medicines are becoming the trademark of cancer treatment; it is a continually evolving approach that is based on the customization of treatments as per the individual genetic makeup. In 2019, the FDA approved 12 personalized medications to investigate and address the root causes of disease, thus combining precision medicine in clinical care.
Threat:
Operational barriers faced in conducting diagnostic tests
Clinical laboratories across significant markets are still developing; technicians face operational challenges in guaranteeing effectual storage, sample procurement, and transportation, particularly while adopting new technologies such as NGS and lab-on-a-chip PCR devices. Laboratory space also wants to be reconfigured to meet the necessities of conducting precise diagnostic tests used for pathogen detection as a means of avoiding cross-contamination and ensuring efficient time management. This results in substantial cost escalation to uphold and operate advanced molecular diagnostic instruments, particularly those capable of handling a single sample type. In addition, owing to the rapid mutation of microbes and the growing outbreak of epidemics, clinical laboratories require to adopt innovative technologies capable of rapid sample diagnosis. Though, the shortage of skilled laboratory technicians to operate advanced diagnostic products has hindered their overall adoption, particularly in emerging markets. Numerous providers find it difficult to transition to IT-based approaches from manual or traditional approaches or may not prefer to make the shift at all.
Covid-19 Impact
As part of the response to COVID 19, virtually all OECD countries affected by the virus have introduced strict restrictions to social and economic life, including social distancing and even full lockdowns. Gaining intelligence on the evolution of the epidemic, including on when a threshold for herd immunity has been reached. In the case of COVID 19, it has been estimated that 50% to 60% of the population needs to be immune to the virus to halt its spread. During the early phase of the coronavirus disease 2019 (COVID-19) pandemic, design, development, validation, verification and implementation of diagnostic tests were actively addressed by a large number of diagnostic test manufacturers. Hundreds of molecular tests and immunoassays were rapidly developed, albeit many still await clinical validation and formal approval.
The molecular diagnostics segment is expected to be the largest during the forecast period
The molecular diagnostics segment is estimated to have a lucrative growth, due to the increasing attention toward the development of genetic therapeutics and growing research in human genomics. Technological advantages in molecular tests make them valuable diagnostic tools for detecting a wide range of genetic diseases. For instance, in November 2017, Edico Genome launched the DRAGEN Clinical Genomics Information System (CGIS). It is designed to enable clinical laboratories to develop sequencing-based laboratory developed tests in a quick, simple, and efficient manner.
The clinical research organizations segment is expected to have the highest CAGR during the forecast period
The clinical research organizations segment is anticipated to witness the fastest CAGR growth during the forecast period, as these organizations can develop LDTs based on any diagnostic test technology used in the clinical lab. For some technologies, such as chemistry tests or immunoassays, automated platforms with large test menus are commercially available. As a result, in some labs, there may be less development and use of LDTs based on these technologies than LDTs based on newer technologies such as next-generation sequencing. Clinical research organizations may also perform tests on types of specimens that are not included in the approved labeling for a test or may have other reasons why they decide to develop an LDT.
Region with highest share:
North America is projected to hold the largest market share during the forecast period, due to the developed laboratory infrastructure, availability of skilled personnel in the region, technological advancements, and trends like personalized medicine, and growing elderly population. The elderly population in countries like the United States are covered with social reimbursement schemes like MediCaid, which provides all healthcare benefits to people over 65. The growth of the elderly population, the cost-effective nature of lab-based testing, and available social support will drive tremendous growth for players in the market.
Region with highest CAGR:
Asia Pacific is projected to have the highest CAGR over the forecast period due to the presence of well-developed healthcare infrastructure, government support, increasing research and development activities along with the growing medical tourism in the region, growing usage of laboratory developed test in combating Covid-19, increasing incidents of cancer and genetic disorders, and large number of product launches. In Asia Pacific, India is the largest market for laboratory developed test.
Key players in the market
Some of the key players profiled in the Laboratory-Developed Testing Market include Qiagen, Epic Sciences, Inc., F. Hoffmann-La Roche Ltd, Bruker Corporation, Illumina, Inc., Bio-Rad Laboratories, Inc., Eurofins Scientific, Thermo Fisher Scientific Inc., Quest Diagnostics, Siemens Healthcare Private Limited, Sonic Healthcare Ltd., Abbott Laboratories, Adaptive Biotechnologies, OPKO Health Inc., Rosetta Genomics Ltd., Guardant Health, Genoptix, Inc., and Quidel Corporation.
Key Developments:
In May 2022, Epic Sciences, Inc. announced the launch of DefineMBC™, a novel comprehensive blood-based test for distinguishing metastatic breast cancer. This test will provide both cell-based and cell-free analysis from a single blood draw to provide comprehensive profiling in the absence of a tissue biopsy result.
In March, 2022, Illumina, Inc. announced the release of TruSightTM Oncology (TSO), a single test that examines numerous tumor genes and biomarkers to determine a patient's cancer molecular profile.
In December 2021, Roche launched the AVENIO Edge System, which simplifies and automates sample preparation for next-generation sequencing, reducing human error and advancing precision medicine.
In September 2021, QIAGEN N.V. received a contract from the United States government to grow its production capacity for consumables for its NeuMoDx 96 and 288 integrated PCR testing systems.
In January 2021, Eurofins launched the NovaType test for detecting and monitoring new variants of SARS-CoV-2. This RT-PCR assay is clinically validated for identifying B.1.1.7 and B.1.351 variants with a short time. NovaType is already available as an LDT in Germany, and it will be made available in over 50 Eurofins laboratories worldwide for testing patients for COVID-19.
Test Types Covered:
• Critical Care
• Microbiology
• Clinical Biochemistry
• Immunology
• Haematology
• Molecular Diagnostics
Products Covered:
• Research and Development (R&D)
• Commercial Service
• Mass Spectrometry
• Microarrays
• Polymerase Chain Reaction (PCR)
• Next-Generation Sequencing (NGS)
Applications Covered:
• Genetics
• Autoimmune Disorders
• Oncology
• Infectious Diseases
• Neurology
End Users Covered:
• Specialty Diagnostics Centers
• Academic Institutes
• Hospital Laboratories
• Clinical Research Organizations
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2020, 2021, 2022, 2025, and 2028
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
o Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Product Analysis
3.7 Application Analysis
3.8 End User Analysis
3.9 Emerging Markets
3.10 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Laboratory-Developed Testing Market, By Test Type
5.1 Introduction
5.2 Critical Care
5.3 Microbiology
5.4 Clinical Biochemistry
5.5 Immunology
5.6 Haematology
5.6.1 Hemoglobin Testing
5.6.2 Coagulation and Hemostasis
5.6.3 Blood Count Testing
5.7 Molecular Diagnostics
6 Global Laboratory-Developed Testing Market, By Product
6.1 Introduction
6.2 Research and Development (R&D)
6.3 Commercial Service
6.4 Mass Spectrometry
6.5 Microarrays
6.6 Polymerase Chain Reaction (PCR)
6.7 Next-Generation Sequencing (NGS)
7 Global Laboratory-Developed Testing Market, By Application
7.1 Introduction
7.2 Genetics
7.3 Autoimmune Disorders
7.4 Oncology
7.5 Infectious Diseases
7.6 Neurology
8 Global Laboratory-Developed Testing Market, By End User
8.1 Introduction
8.2 Specialty Diagnostics Centers
8.3 Academic Institutes
8.4 Hospital Laboratories
8.5 Clinical Research Organizations
9 Global Laboratory-Developed Testing Market, By Geography
9.1 Introduction
9.2 North America
9.2.1 US
9.2.2 Canada
9.2.3 Mexico
9.3 Europe
9.3.1 Germany
9.3.2 UK
9.3.3 Italy
9.3.4 France
9.3.5 Spain
9.3.6 Rest of Europe
9.4 Asia Pacific
9.4.1 Japan
9.4.2 China
9.4.3 India
9.4.4 Australia
9.4.5 New Zealand
9.4.6 South Korea
9.4.7 Rest of Asia Pacific
9.5 South America
9.5.1 Argentina
9.5.2 Brazil
9.5.3 Chile
9.5.4 Rest of South America
9.6 Middle East & Africa
9.6.1 Saudi Arabia
9.6.2 UAE
9.6.3 Qatar
9.6.4 South Africa
9.6.5 Rest of Middle East & Africa
10 Key Developments
10.1 Agreements, Partnerships, Collaborations and Joint Ventures
10.2 Acquisitions & Mergers
10.3 New Product Launch
10.4 Expansions
10.5 Other Key Strategies
11 Company Profiling
11.1 Qiagen
11.2 Epic Sciences, Inc.
11.3 F. Hoffmann-La Roche Ltd
11.4 Bruker Corporation
11.5 Illumina, Inc.
11.6 Bio-Rad Laboratories, Inc.
11.7 Eurofins Scientific
11.8 Thermo Fisher Scientific Inc.
11.9 Quest Diagnostics
11.10 Siemens Healthcare Private Limited
11.11 Sonic Healthcare Ltd.
11.12 Abbott Laboratories
11.13 Adaptive Biotechnologies
11.14 OPKO Health Inc.
11.15 Rosetta Genomics Ltd.
11.16 Guardant Health
11.17 Genoptix, Inc.
11.18 Quidel Corporation
List of Tables
1 Global Laboratory-Developed Testing Market Outlook, By Region (2020-2028) ($MN)
2 Global Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
3 Global Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
4 Global Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
5 Global Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
6 Global Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
7 Global Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
8 Global Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
9 Global Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
10 Global Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
11 Global Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
12 Global Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
13 Global Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
14 Global Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
15 Global Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
16 Global Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
17 Global Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
18 Global Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
19 Global Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
20 Global Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
21 Global Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
22 Global Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
23 Global Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
24 Global Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
25 Global Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
26 Global Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
27 Global Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
28 Global Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
29 Global Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
30 North America Laboratory-Developed Testing Market Outlook, By Country (2020-2028) ($MN)
31 North America Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
32 North America Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
33 North America Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
34 North America Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
35 North America Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
36 North America Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
37 North America Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
38 North America Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
39 North America Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
40 North America Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
41 North America Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
42 North America Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
43 North America Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
44 North America Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
45 North America Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
46 North America Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
47 North America Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
48 North America Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
49 North America Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
50 North America Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
51 North America Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
52 North America Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
53 North America Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
54 North America Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
55 North America Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
56 North America Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
57 North America Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
58 North America Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
59 Europe Laboratory-Developed Testing Market Outlook, By Country (2020-2028) ($MN)
60 Europe Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
61 Europe Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
62 Europe Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
63 Europe Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
64 Europe Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
65 Europe Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
66 Europe Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
67 Europe Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
68 Europe Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
69 Europe Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
70 Europe Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
71 Europe Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
72 Europe Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
73 Europe Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
74 Europe Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
75 Europe Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
76 Europe Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
77 Europe Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
78 Europe Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
79 Europe Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
80 Europe Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
81 Europe Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
82 Europe Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
83 Europe Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
84 Europe Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
85 Europe Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
86 Europe Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
87 Europe Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
88 Asia Pacific Laboratory-Developed Testing Market Outlook, By Country (2020-2028) ($MN)
89 Asia Pacific Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
90 Asia Pacific Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
91 Asia Pacific Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
92 Asia Pacific Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
93 Asia Pacific Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
94 Asia Pacific Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
95 Asia Pacific Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
96 Asia Pacific Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
97 Asia Pacific Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
98 Asia Pacific Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
99 Asia Pacific Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
100 Asia Pacific Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
101 Asia Pacific Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
102 Asia Pacific Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
103 Asia Pacific Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
104 Asia Pacific Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
105 Asia Pacific Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
106 Asia Pacific Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
107 Asia Pacific Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
108 Asia Pacific Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
109 Asia Pacific Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
110 Asia Pacific Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
111 Asia Pacific Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
112 Asia Pacific Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
113 Asia Pacific Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
114 Asia Pacific Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
115 Asia Pacific Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
116 Asia Pacific Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
117 South America Laboratory-Developed Testing Market Outlook, By Country (2020-2028) ($MN)
118 South America Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
119 South America Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
120 South America Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
121 South America Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
122 South America Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
123 South America Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
124 South America Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
125 South America Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
126 South America Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
127 South America Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
128 South America Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
129 South America Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
130 South America Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
131 South America Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
132 South America Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
133 South America Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
134 South America Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
135 South America Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
136 South America Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
137 South America Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
138 South America Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
139 South America Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
140 South America Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
141 South America Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
142 South America Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
143 South America Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
144 South America Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
145 South America Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
146 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Country (2020-2028) ($MN)
147 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Test Type (2020-2028) ($MN)
148 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Critical Care (2020-2028) ($MN)
149 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Microbiology (2020-2028) ($MN)
150 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Clinical Biochemistry (2020-2028) ($MN)
151 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Immunology (2020-2028) ($MN)
152 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Haematology (2020-2028) ($MN)
153 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Hemoglobin Testing (2020-2028) ($MN)
154 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Coagulation and Hemostasis (2020-2028) ($MN)
155 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Blood Count Testing (2020-2028) ($MN)
156 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Molecular Diagnostics (2020-2028) ($MN)
157 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Product (2020-2028) ($MN)
158 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Research and Development (R&D) (2020-2028) ($MN)
159 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Commercial Service (2020-2028) ($MN)
160 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Mass Spectrometry (2020-2028) ($MN)
161 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Microarrays (2020-2028) ($MN)
162 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Polymerase Chain Reaction (PCR) (2020-2028) ($MN)
163 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Next-Generation Sequencing (NGS) (2020-2028) ($MN)
164 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Application (2020-2028) ($MN)
165 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Genetics (2020-2028) ($MN)
166 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Autoimmune Disorders (2020-2028) ($MN)
167 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Oncology (2020-2028) ($MN)
168 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Infectious Diseases (2020-2028) ($MN)
169 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Neurology (2020-2028) ($MN)
170 Middle East & Africa Laboratory-Developed Testing Market Outlook, By End User (2020-2028) ($MN)
171 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Specialty Diagnostics Centers (2020-2028) ($MN)
172 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Academic Institutes (2020-2028) ($MN)
173 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Hospital Laboratories (2020-2028) ($MN)
174 Middle East & Africa Laboratory-Developed Testing Market Outlook, By Clinical Research Organizations (2020-2028) ($MN)
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We provide a free 15% customization on every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
Note: This customization is absolutely free until it falls under the 15% bracket. If your requirement exceeds this a feasibility check will be performed. Post that, a quote will be provided along with the timelines.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
