
Clinical Trial Packaging Market
Clinical Trial Packaging Market Forecasts to 2030 - Global Analysis By Product Type (Syringes, Vials & Ampoules, Bottles, Bags & Pouches, Blisters, Tubes, Sachets, Kits or Packs and Other Product Types), Clinical Trial Type (Therapeutic and Prevention, Diagnostics and Other Clinical Trail Types), Material (Plastic, Glass, Metal, Paper, Corrugated Fiber and Other Materials), End User and By Geography

|
Years Covered |
2021-2030 |
|
Estimated Year Value (2023) |
US $3.04 BN |
|
Projected Year Value (2030) |
US $7.03 BN |
|
CAGR (2023 - 2030) |
12.7% |
|
Regions Covered |
North America, Europe, Asia Pacific, South America, and Middle East & Africa |
|
Countries Covered |
US, Canada, Mexico, Germany, UK, Italy, France, Spain, Japan, China, India, Australia, New Zealand, South Korea, Rest of Asia Pacific, South America, Argentina, Brazil, Chile, Middle East & Africa, Saudi Arabia, UAE, Qatar, and South Africa |
|
Largest Market |
North America |
|
Highest Growing Market |
Asia Pacific |
According to Stratistics MRC, the Global Clinical Trial Packaging Market is accounted for $3.04 billion in 2023 and is expected to reach $7.03 billion by 2030 growing at a CAGR of 12.7% during the forecast period. Clinical trial products are packaged, stored, protected, and transported using clinical trial packaging solutions. Clinical trials are conducted to evaluate a drug's effectiveness and determine the best dose for various pharmaceutical formulations. Clinical trials involve the use of a wide range of products, including syringes, bottles, blisters, tubes, ampoules and vials, bags and pouches, and sachets. Moreover, these products require special packaging because standard packaging has a negative impact on clinical trial outcomes, particularly for highly sensitive drugs.
According to CPSC, the options available to us must incorporate physician involvement in assessing dispensation of drug to individual patients.
Market Dynamics:
Driver:
Revolution in biologics and personalized medicine
With the development of biologics like monoclonal antibodies, cell therapies, and gene therapies, the field of clinical trials has undergone significant change. To maintain their stability and integrity, these cutting-edge therapies frequently need specialized packaging solutions. Additionally, the packaging requirements have become even more varied as a result of personalized medicine, which adapts treatments to specific patients. To accommodate various dosages, formulations, and administration techniques, specialized packaging solutions are required.
Restraint:
Environmental concerns with sustainability
A significant barrier to the market for clinical trial packaging is the increasing focus on environmental sustainability. In order to lessen their impact on the environment, businesses are under increasing pressure to adopt eco-friendly packaging strategies. Furthermore, the cost of developing and implementing these environmentally friendly packaging options may be higher, which will have an impact on profit margins and operating expenses even though they are in line with global environmental goals.
Opportunity:
Integration of digital technologies
Clinical trial packaging offers significant opportunities when digital technologies are incorporated. Innovations like blockchain for secure data sharing, QR codes for improved traceability, and IoT-enabled packaging with real-time monitoring capabilities are revolutionizing the packaging industry. Moreover, packaging companies that use these technologies can improve supply chains for clinical trials by doing so while lowering errors and increasing efficiency.
Threat:
Rising costs of raw materials
The clinical trial packaging market is constantly threatened by changes in the price of raw materials like plastics, metals, and specialty packaging components. Sudden price increases can reduce profit margins, which affects how cost-effective packaging solutions are overall. Additionally, companies need to establish effective procurement strategies and closely monitor the markets for raw materials to reduce this threat. This will help them control cost fluctuations.
Covid-19 Impact:
The clinical trial packaging market was significantly affected by the COVID-19 pandemic. Initial delays and shortages in packaging supplies had an impact on the timely completion of clinical trials due to disruptions in global supply chains and manufacturing procedures. Furthermore, the distribution of clinical trial materials was hampered by strict lockdown procedures and travel restrictions, creating logistical difficulties. But the pandemic also sped up industry wide efforts to transform digitally, leading to the adoption of remote monitoring and smart packaging technologies.
The Bottles segment is expected to be the largest during the forecast period
During the forecast period, it is anticipated that the bottle segment will hold the largest share. Many different types of medications, including oral medications, liquids, and even solid dosage forms like tablets and capsules, are packaged in bottles. They are adaptable, simple to use, and can accommodate various storage and dispensing requirements. Additionally, bottles are available in a range of sizes and materials, so pharmaceutical companies can select packaging options that meet the particular needs of their products.
The Clinical Research Organizations segment is expected to have the highest CAGR during the forecast period
In the market for clinical trial packaging, the clinical research organization segment is growing at the highest CAGR during the anticipated period. For the benefit of pharmaceutical and biotechnology companies, CROs are essential in carrying out clinical trials. They manage a sizable portion of clinical research tasks, such as patient recruitment, data management, and legal compliance. Furthermore, Pharmaceutical and biotechnology companies are increasingly outsourcing clinical trial activities to CROs due to cost savings and specialized knowledge, which is fueling these companies' remarkable growth. The growing number of clinical trials and the increase in demand for clinical trial packaging services also contributed to the growth of the CRO segment.
Region with largest share:
The largest market share for clinical trial packaging was held by North America, particularly the United States. North America's dominance can be attributed to its thriving biotechnology and pharmaceutical industries, extensive clinical trial activities, well-established regulatory framework, and substantial investments in research and development. Moreover, this region leads the world in clinical trial packaging due to its sophisticated healthcare system and commitment to innovation.
Region with highest CAGR:
In the market for clinical trial packaging, the Asia-Pacific region is expected to have the highest CAGR. The region's large and diverse patient population and the burgeoning pharmaceutical and biotechnology industries were the main drivers of this growth, as were raising investments in healthcare infrastructure. Additionally, some Asian nation’s affordable manufacturing and packaging options added to the region's appeal as a source of clinical trial packaging services.
Key players in the market
Some of the key players in Clinical Trial Packaging market include PCI Pharma Services, Schreiner MediPharm, Berry Global, Inc., 3M Company, Nupharm Group , Catalent, Inc. , Sharp Clinical Services, Inc. , Körber Medipak Systems AG, Parexel International Corporation , Alcami Corporation, Inc., Reed-Lane, Inc., Aphena Pharma Solutions, Symbiosis Pharmaceutical Services, Carton Service, Inc., Piramal Pharma Solutions , Thermo Fisher Scientific Inc., Xerimis Inc., NextPharma Technologies Holding Limited, WestRock Company, Sentry BioPharma Services, Inc. and Yourway Transport Inc.
Key Developments:
In May 2023, 3M today announced it has entered into agreements to sell certain assets associated with its dental local anesthetic portfolio, based in Seefeld, Germany, to Pierrel S.p.A., a global provider of services for the pharmaceutical industry, for a purchase price of $70 million, subject to closing and other adjustments.
In May 2023, Schreiner MediPharm has introduced a counterfeit and tampering protection solution whose void effect makes it exceedingly difficult for counterfeiters to successfully reuse original packaging. The company’s tailored Void-Labels make previously covert messages readily visible upon peeling, making a package’s first opening clearly and irreversibly apparent.
In April 2023, PCI Pharma Services (PCI), a leading global contract development and manufacturing organization announced that three new state-of-the-arts automated sterile fill-finish machines at its San Diego and Melbourne facilities are now fully operational. The innovative machinery from Cytiva can be used to fill various sterile medications into vials and syringes for small-to-mid scale client needs.
Product Types Covered:
• Syringes
• Vials & Ampoules
• Bottles
• Bags & Pouches
• Blisters
• Tubes
• Sachets
• Kits or Packs
• Other Product Types
Clinical Trial Types Covered:
• Therapeutic and Prevention
• Diagnostics
• Other Clinical Trail Types
Materials Covered:
• Plastic
• Glass
• Metal
• Paper
• Corrugated Fiber
• Other Materials
End Users Covered:
• Research Laboratories
• Clinical Research Organizations
• Drug Manufacturing Facilities
• Pharmaceutical Companies
• Biotechnology Companies
• Academic & Research Institutes
• Hospitals & Clinics
• Other End Users
Regions Covered:
• North America
o US
o Canada
o Mexico
• Europe
o Germany
o UK
o Italy
o France
o Spain
o Rest of Europe
• Asia Pacific
o Japan
o China
o India
o Australia
o New Zealand
o South Korea
o Rest of Asia Pacific
• South America
o Argentina
o Brazil
o Chile
o Rest of South America
• Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Rest of Middle East & Africa
What our report offers:
- Market share assessments for the regional and country-level segments
- Strategic recommendations for the new entrants
- Covers Market data for the years 2021, 2022, 2023, 2026, and 2030
- Market Trends (Drivers, Constraints, Opportunities, Threats, Challenges, Investment Opportunities, and recommendations)
- Strategic recommendations in key business segments based on the market estimations
- Competitive landscaping mapping the key common trends
- Company profiling with detailed strategies, financials, and recent developments
- Supply chain trends mapping the latest technological advancements
Free Customization Offerings:
All the customers of this report will be entitled to receive one of the following free customization options:
• Company Profiling
o Comprehensive profiling of additional market players (up to 3)
o SWOT Analysis of key players (up to 3)
• Regional Segmentation
o Market estimations, Forecasts and CAGR of any prominent country as per the client's interest (Note: Depends on feasibility check)
• Competitive Benchmarking
Benchmarking of key players based on product portfolio, geographical presence, and strategic alliances
Table of Contents
1 Executive Summary
2 Preface
2.1 Abstract
2.2 Stake Holders
2.3 Research Scope
2.4 Research Methodology
2.4.1 Data Mining
2.4.2 Data Analysis
2.4.3 Data Validation
2.4.4 Research Approach
2.5 Research Sources
2.5.1 Primary Research Sources
2.5.2 Secondary Research Sources
2.5.3 Assumptions
3 Market Trend Analysis
3.1 Introduction
3.2 Drivers
3.3 Restraints
3.4 Opportunities
3.5 Threats
3.6 Product Analysis
3.7 End User Analysis
3.8 Emerging Markets
3.9 Impact of Covid-19
4 Porters Five Force Analysis
4.1 Bargaining power of suppliers
4.2 Bargaining power of buyers
4.3 Threat of substitutes
4.4 Threat of new entrants
4.5 Competitive rivalry
5 Global Clinical Trial Packaging Market, By Product Type
5.1 Introduction
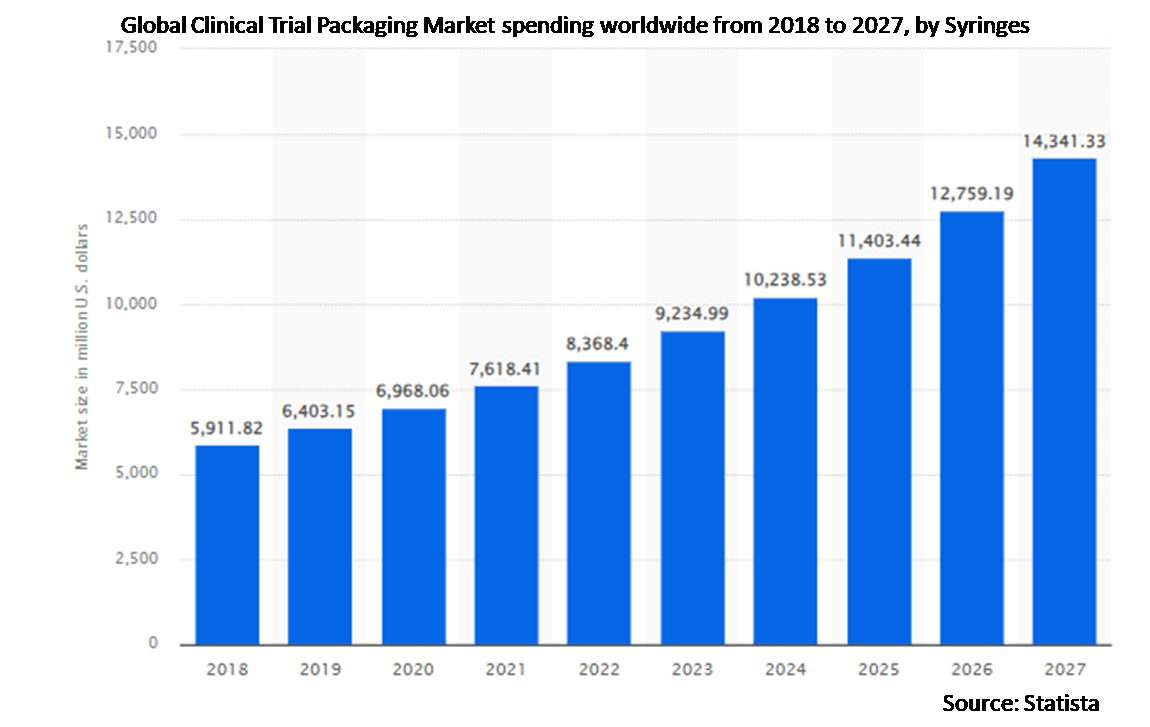
5.2 Syringes
5.3 Vials & Ampoules
5.4 Bottles
5.5 Bags & Pouches
5.6 Blisters
5.6.1 Cold Forming
5.6.2 Thermoforming
5.7 Tubes
5.8 Sachets
5.9 Kits or Packs
5.10 Other Product Types
6 Global Clinical Trial Packaging Market, By Clinical Trial Type
6.1 Introduction
6.2 Therapeutic and Prevention
6.2.1 Vaccines
6.2.2 Drug Discovery and Development
6.2.3 Therapeutic Devices
6.2.4 Biosimilars
6.2.5 Therapeutic Assays
6.2.6 Therapeutic Procedures
6.3 Diagnostics
6.3.1 Diagnostic Assay
6.3.2 Diagnostic Devices
6.4 Other Clinical Trail Types
7 Global Clinical Trial Packaging Market, By Material
7.1 Introduction
7.2 Plastic
7.2.1 Polyvinyl Chloride (PVC)
7.2.2 Polyethylene (PE)
7.2.2.1 High Density Polyethylene (HDPE)
7.2.2.2 Low Density Polyethylene (LDPE)
7.2.3 Polypropylene (PP)
7.3 Glass
7.4 Metal
7.5 Paper
7.6 Corrugated Fiber
7.7 Other Materials
8 Global Clinical Trial Packaging Market, By End User
8.1 Introduction
8.2 Research Laboratories
8.3 Clinical Research Organizations
8.4 Drug Manufacturing Facilities
8.5 Pharmaceutical Companies
8.6 Biotechnology Companies
8.7 Academic & Research Institutes
8.8 Hospitals & Clinics
8.9 Other End Users
9 Global Clinical Trial Packaging Market, By Geography
9.1 Introduction
9.2 North America
9.2.1 US
9.2.2 Canada
9.2.3 Mexico
9.3 Europe
9.3.1 Germany
9.3.2 UK
9.3.3 Italy
9.3.4 France
9.3.5 Spain
9.3.6 Rest of Europe
9.4 Asia Pacific
9.4.1 Japan
9.4.2 China
9.4.3 India
9.4.4 Australia
9.4.5 New Zealand
9.4.6 South Korea
9.4.7 Rest of Asia Pacific
9.5 South America
9.5.1 Argentina
9.5.2 Brazil
9.5.3 Chile
9.5.4 Rest of South America
9.6 Middle East & Africa
9.6.1 Saudi Arabia
9.6.2 UAE
9.6.3 Qatar
9.6.4 South Africa
9.6.5 Rest of Middle East & Africa
10 Key Developments
10.1 Agreements, Partnerships, Collaborations and Joint Ventures
10.2 Acquisitions & Mergers
10.3 New Product Launch
10.4 Expansions
10.5 Other Key Strategies
11 Company Profiling
11.1 PCI Pharma Services
11.2 Schreiner MediPharm
11.3 Berry Global, Inc.
11.4 3M Company
11.5 Nupharm Group
11.6 Catalent, Inc.
11.7 Sharp Clinical Services, Inc.
11.8 Körber Medipak Systems AG
11.9 Parexel International Corporation
11.10 Alcami Corporation, Inc.
11.11 Reed-Lane, Inc.
11.12 Aphena Pharma Solutions
11.13 Symbiosis Pharmaceutical Services
11.14 Carton Service, Inc.
11.15 Piramal Pharma Solutions
11.16 Thermo Fisher Scientific Inc.
11.17 Xerimis Inc.
11.18 NextPharma Technologies Holding Limited
11.19 WestRock Company
11.20 Sentry BioPharma Services, Inc.
11.21 Yourway Transport Inc.
List of Tables
1 Global Clinical Trial Packaging Market Outlook, By Region (2021-2030) ($MN)
2 Global Clinical Trial Packaging Market Outlook, By Product Type (2021-2030) ($MN)
3 Global Clinical Trial Packaging Market Outlook, By Syringes (2021-2030) ($MN)
4 Global Clinical Trial Packaging Market Outlook, By Vials & Ampoules (2021-2030) ($MN)
5 Global Clinical Trial Packaging Market Outlook, By Bottles (2021-2030) ($MN)
6 Global Clinical Trial Packaging Market Outlook, By Bags & Pouches (2021-2030) ($MN)
7 Global Clinical Trial Packaging Market Outlook, By Blisters (2021-2030) ($MN)
8 Global Clinical Trial Packaging Market Outlook, By Cold Forming (2021-2030) ($MN)
9 Global Clinical Trial Packaging Market Outlook, By Thermoforming (2021-2030) ($MN)
10 Global Clinical Trial Packaging Market Outlook, By Tubes (2021-2030) ($MN)
11 Global Clinical Trial Packaging Market Outlook, By Sachets (2021-2030) ($MN)
12 Global Clinical Trial Packaging Market Outlook, By Kits or Packs (2021-2030) ($MN)
13 Global Clinical Trial Packaging Market Outlook, By Other Product Types (2021-2030) ($MN)
14 Global Clinical Trial Packaging Market Outlook, By Clinical Trial Type (2021-2030) ($MN)
15 Global Clinical Trial Packaging Market Outlook, By Therapeutic and Prevention (2021-2030) ($MN)
16 Global Clinical Trial Packaging Market Outlook, By Vaccines (2021-2030) ($MN)
17 Global Clinical Trial Packaging Market Outlook, By Drug Discovery and Development (2021-2030) ($MN)
18 Global Clinical Trial Packaging Market Outlook, By Therapeutic Devices (2021-2030) ($MN)
19 Global Clinical Trial Packaging Market Outlook, By Biosimilars (2021-2030) ($MN)
20 Global Clinical Trial Packaging Market Outlook, By Therapeutic Assays (2021-2030) ($MN)
21 Global Clinical Trial Packaging Market Outlook, By Therapeutic Procedures (2021-2030) ($MN)
22 Global Clinical Trial Packaging Market Outlook, By Diagnostics (2021-2030) ($MN)
23 Global Clinical Trial Packaging Market Outlook, By Diagnostic Assay (2021-2030) ($MN)
24 Global Clinical Trial Packaging Market Outlook, By Diagnostic Devices (2021-2030) ($MN)
25 Global Clinical Trial Packaging Market Outlook, By Other Clinical Trail Types (2021-2030) ($MN)
26 Global Clinical Trial Packaging Market Outlook, By Material (2021-2030) ($MN)
27 Global Clinical Trial Packaging Market Outlook, By Plastic (2021-2030) ($MN)
28 Global Clinical Trial Packaging Market Outlook, By Polyvinyl Chloride (PVC) (2021-2030) ($MN)
29 Global Clinical Trial Packaging Market Outlook, By Polyethylene (PE) (2021-2030) ($MN)
30 Global Clinical Trial Packaging Market Outlook, By Polypropylene (PP) (2021-2030) ($MN)
31 Global Clinical Trial Packaging Market Outlook, By Glass (2021-2030) ($MN)
32 Global Clinical Trial Packaging Market Outlook, By Metal (2021-2030) ($MN)
33 Global Clinical Trial Packaging Market Outlook, By Paper (2021-2030) ($MN)
34 Global Clinical Trial Packaging Market Outlook, By Corrugated Fiber (2021-2030) ($MN)
35 Global Clinical Trial Packaging Market Outlook, By Other Materials (2021-2030) ($MN)
36 Global Clinical Trial Packaging Market Outlook, By End User (2021-2030) ($MN)
37 Global Clinical Trial Packaging Market Outlook, By Research Laboratories (2021-2030) ($MN)
38 Global Clinical Trial Packaging Market Outlook, By Clinical Research Organizations (2021-2030) ($MN)
39 Global Clinical Trial Packaging Market Outlook, By Drug Manufacturing Facilities (2021-2030) ($MN)
40 Global Clinical Trial Packaging Market Outlook, By Pharmaceutical Companies (2021-2030) ($MN)
41 Global Clinical Trial Packaging Market Outlook, By Biotechnology Companies (2021-2030) ($MN)
42 Global Clinical Trial Packaging Market Outlook, By Academic & Research Institutes (2021-2030) ($MN)
43 Global Clinical Trial Packaging Market Outlook, By Hospitals & Clinics (2021-2030) ($MN)
44 Global Clinical Trial Packaging Market Outlook, By Other End Users (2021-2030) ($MN)
Note: Tables for North America, Europe, APAC, South America, and Middle East & Africa Regions are also represented in the same manner as above.
List of Figures
RESEARCH METHODOLOGY

We at ‘Stratistics’ opt for an extensive research approach which involves data mining, data validation, and data analysis. The various research sources include in-house repository, secondary research, competitor’s sources, social media research, client internal data, and primary research.
Our team of analysts prefers the most reliable and authenticated data sources in order to perform the comprehensive literature search. With access to most of the authenticated data bases our team highly considers the best mix of information through various sources to obtain extensive and accurate analysis.
Each report takes an average time of a month and a team of 4 industry analysts. The time may vary depending on the scope and data availability of the desired market report. The various parameters used in the market assessment are standardized in order to enhance the data accuracy.
Data Mining
The data is collected from several authenticated, reliable, paid and unpaid sources and is filtered depending on the scope & objective of the research. Our reports repository acts as an added advantage in this procedure. Data gathering from the raw material suppliers, distributors and the manufacturers is performed on a regular basis, this helps in the comprehensive understanding of the products value chain. Apart from the above mentioned sources the data is also collected from the industry consultants to ensure the objective of the study is in the right direction.
Market trends such as technological advancements, regulatory affairs, market dynamics (Drivers, Restraints, Opportunities and Challenges) are obtained from scientific journals, market related national & international associations and organizations.
Data Analysis
From the data that is collected depending on the scope & objective of the research the data is subjected for the analysis. The critical steps that we follow for the data analysis include:
- Product Lifecycle Analysis
- Competitor analysis
- Risk analysis
- Porters Analysis
- PESTEL Analysis
- SWOT Analysis
The data engineering is performed by the core industry experts considering both the Marketing Mix Modeling and the Demand Forecasting. The marketing mix modeling makes use of multiple-regression techniques to predict the optimal mix of marketing variables. Regression factor is based on a number of variables and how they relate to an outcome such as sales or profits.
Data Validation
The data validation is performed by the exhaustive primary research from the expert interviews. This includes telephonic interviews, focus groups, face to face interviews, and questionnaires to validate our research from all aspects. The industry experts we approach come from the leading firms, involved in the supply chain ranging from the suppliers, distributors to the manufacturers and consumers so as to ensure an unbiased analysis.
We are in touch with more than 15,000 industry experts with the right mix of consultants, CEO's, presidents, vice presidents, managers, experts from both supply side and demand side, executives and so on.
The data validation involves the primary research from the industry experts belonging to:
- Leading Companies
- Suppliers & Distributors
- Manufacturers
- Consumers
- Industry/Strategic Consultants
Apart from the data validation the primary research also helps in performing the fill gap research, i.e. providing solutions for the unmet needs of the research which helps in enhancing the reports quality.
For more details about research methodology, kindly write to us at info@strategymrc.com
Frequently Asked Questions
In case of any queries regarding this report, you can contact the customer service by filing the “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
Yes, the samples are available for all the published reports. You can request them by filling the “Request Sample” option available in this page.
Yes, you can request a sample with your specific requirements. All the customized samples will be provided as per the requirement with the real data masked.
All our reports are available in Digital PDF format. In case if you require them in any other formats, such as PPT, Excel etc you can submit a request through “Inquiry Before Buy” form available on the right hand side. You may also contact us through email: info@strategymrc.com or phone: +1-301-202-5929
We offer a free 15% customization with every purchase. This requirement can be fulfilled for both pre and post sale. You may send your customization requirements through email at info@strategymrc.com or call us on +1-301-202-5929.
We have 3 different licensing options available in electronic format.
- Single User Licence: Allows one person, typically the buyer, to have access to the ordered product. The ordered product cannot be distributed to anyone else.
- 2-5 User Licence: Allows the ordered product to be shared among a maximum of 5 people within your organisation.
- Corporate License: Allows the product to be shared among all employees of your organisation regardless of their geographical location.
All our reports are typically be emailed to you as an attachment.
To order any available report you need to register on our website. The payment can be made either through CCAvenue or PayPal payments gateways which accept all international cards.
We extend our support to 6 months post sale. A post sale customization is also provided to cover your unmet needs in the report.
Request Customization
We offer complimentary customization of up to 15% with every purchase. To share your customization requirements, feel free to email us at info@strategymrc.com or call us on +1-301-202-5929. .
Please Note: Customization within the 15% threshold is entirely free of charge. If your request exceeds this limit, we will conduct a feasibility assessment. Following that, a detailed quote and timeline will be provided.
WHY CHOOSE US ?

Assured Quality
Best in class reports with high standard of research integrity

24X7 Research Support
Continuous support to ensure the best customer experience.

Free Customization
Adding more values to your product of interest.

Safe & Secure Access
Providing a secured environment for all online transactions.

Trusted by 600+ Brands
Serving the most reputed brands across the world.
